|
| |
Groundwork
Colorado Rocks
You Name It, We've Got It
|

|
|
|
 Under
construction Under
construction
Last modified 10/22/04
Top
Page Index
Introduction
When I first thought to name this article
"Colorado Rocks", my hope was to limit its scope. But in truth, it's
hard to name a rock Colorado doesn't have. Some of the higher-grade metamorphic
rocks, like the 1.7 Ga arc-related
metavolcanics, may bear little resemblance to their original state, but in one form or
another, they're all here.
Petrology
Petrology, the study of rocks, may sound
absurd to some, but it's more practical than it sounds. Rocks hold the
only available record of the history of our one and only planet. They also hold
the key to two of the great pillars of human economy—mineral wealth and
agricultural vigor. They tend to hold up the planet's most inspiring scenery,
and they ultimately support everything we build—including houses, schools,
skyscrapers, roads, bridges, tunnels and nuclear power plants.
Our biological history is also more entwined with rocks than you
might think. Cell biologists studying the origins of life now have good evidence
that the precursors of modern cells used rock surfaces as both cell membranes
and as as catalysts for the organic reactions they required. The chimney-like mid-ocean
ridge hydrothermal vents known as ^black
smokers carry on such such rock-cell partnerships to this day. It's no accident
that many
important human enzymes and physiologically active proteins require metal ions as ^co-factors—e.g.,
iron in hemoglobin, magnesium in chlorophyll, chromium in insulin, copper
in cytochrome c oxidase, and zinc in angiotensin converting enzyme, to name just
a few.
Since mantle
and lower crust rocks are only rarely
exposed, the rocks of the upper crust are
the main focus of petrology, even though they constitute well under 1% of
the earth by volume. Of course, geologists are eager to study any rock they
can get their hands on, so they particularly prize the occasional plums
thrust up to the surface from lower levels.
Available Elements
Since the ^chemical
elements are the fundamental building blocks of all ordinary materials, including
minerals, let's start there. Elemental abundances at and near the surface of the earth tightly constrain
the range of minerals and rocks observed, not to mention the range of possible
biologic processes.
The table below lists the 14 most
abundant elements in the earth's crust in decreasing order.
Abundances of Major and Minor Elements in the Earth's
Crust
|
|
Group |
Element |
Chemical Symbol |
Atomic Weight |
Abundance (wt. %) |
|
Major Elements |
Oxygen |
O |
16 |
46.6 |
|
Silicon |
Si |
28 |
27.7 |
|
Aluminum |
Al |
27 |
8.1 |
|
Iron |
Fe |
56 |
5.0 |
| Calcium |
Ca |
40 |
3.6 |
| Sodium |
Na |
23 |
2.8 |
| Potassium |
K |
39 |
2.6 |
|
Magnesium |
Mg |
24 |
2.1 |
|
Subtotal |
98.5 |
| Minor
Elements |
Titanium |
Ti |
48 |
0.4 |
| Hydrogen |
H |
1 |
0.1 |
| Phosphorus |
P |
31 |
0.1 |
| Manganese |
Mn |
55 |
0.1 |
| Sulfur |
S |
32 |
0.05 |
|
Carbon |
C |
12 |
0.03 |
|
Total |
99.3 |
With oxygen and silicon alone accounting for ~74% of
the crust and aluminum for another ~8%, it's little wonder minerals composed
primarily of these three elements dominate the
crust. They do so primarily in the form of silicate and aluminosilicate minerals built on
strong chains and sheets of tetrahedral and octahedral arrays of Si-O and Al-OH bonds.
In nearly all common silicate minerals, including feldspars, micas and clays, positive
ions of calcium, sodium, potassium and magnesium serve both to balance out the negatively
charged silicate backbones and to bind them together neatly without disturbing their crystalline
structures.
It takes seven of the eight major crustal elements
to fill these vital chemical roles in the sialic (Si- and Al-rich) rocks
typical of the upper continental crust.
Iron, the odd atom out in the major element group, figures more prominently in
the higher-density minerals inhabiting the lower continental
crust, the oceanic crust and the mantle.
Olivine, pyroxene, hornblende and biotite are among the most common of these ferromagnesian
or mafic (Ma- and Fe-rich)
minerals.
Trace Elements
Elements beyond the top 14 listed above
fall into the trace element category. They're scarce in the crust for a
very simple reason: By virtue of their size or charge distribution (see below), they fit poorly into the crystalline structures of
typical of most crustal
(silicate) minerals. The trace elements include gold, silver, copper, nickel,
zinc, lead, lithium, beryllium, niobium, tantalum, tin, uranium, thorium, tungsten,
zirconium and the rare earths. Many are more
abundant in the mantle than in the crust. Mantle-derived, water-rich
magmas are their primary means of transport to extractable crustal depths.
New Insights From a
New Periodic Table
In 2003, geochemist Bruce Railsback published his
revolutionary and ingeniously reorganized ^Earth
scientist's periodic table of the elements and their ions showing not only
the neutral elements but also their naturally-occurring ions. Since ions are
with rare exception the stuff of earth materials, much can be learned from their
habits and proclivities. Indeed, since oxygen is by far the most abundant element in
both the mantle and crust, the way various cations (positively charged
ions) interact with ionic oxygen constrains a great deal of geochemistry and to
some extent biochemistry as well. Silicon, the 2nd most abundant element in the crust,
also plays
a defining role in geochemistry.
Ionic Potentials of
Cations
One of the most important innovations in Railsback's
periodic table is the addition of contour lines of equal ionic potential—the ratio z/r of ionic
charge to radius. The higher the ionic potential, the more compact or intense
the ionic electric field, and the more strongly the ion interacts with nearby
charge centers. Since trends in ionic abundance, mineral formation, oxide
melting point, solubility, and even nutrient value all tend to follow contours
of ionic potential, the new table shows at glance important chemical relationships that the
standard table obscures.
Cations (positively charged ions) of low ionic potential (z/r < 4) like Na+,
K+ and Ca2+ bond relatively weakly to O-2, do not form stable
oxide minerals, remain in fluid phases until late in melt
evolution, are highly concentrated and soluble in natural waters and serve as essential nutrients to
both plants and animals.
Cations of intermediate ionic potential (z/r = 3-10)
like Al3+, Fe3+ and Ti4+ bond strongly to O-2.
Their compact and largely shielded charge distributions allow them to coordinate
with a single negative charge center in large numbers with little mutual
repulsion. Such cations tend to form stable oxide minerals, to bond in igneous
minerals early in melt evolution, to concentrate in soil, to linger in the
mantle, to have low concentrations and solubilities in natural waters, to
collect in ferromanganese nodules on the ocean floor, and to serve
inconsequential roles as nutrients.
Cations of high ionic potential (z/r > 8) like P+5,
N+5 and S+6 also bond tightly to O-2 to form
highly stable and soluble radicals like PO4-3,
NO3- and SO4-2, but they can't form
stable oxide minerals due to mutual repulsion. However, the small C+4
cation (z/r ~ 27) forms the stable oxide and greenhouse gas CO2 as
well as stable carbonate oxysalts of the soluble radical CO3-2.
The C+4 cation thus plays a very special role in the planet's surface
temperature-regulating carbonate cycle. High
potential cations share many properties with the cations of low ionic potential. Because they
both readily leach out of soils due to high solubility, K+ (low
potential) and NO3- (high potential) are both key
ingredients in fertilizers.
The most common silicon ion, Si4+,
occupies another special niche as a highly abundant cation at the cusp
(z/r = 8) between high and intermediate ionic potentials. Thanks to similar
ionic potentials, Si4+, V5+, Mo6+ and Se4+
all stand at the upper margin of cations forming stable soluble oxysalts—e.g.,
silicate, SiO4-4 or Si(OH)4—that also form stable
insoluble oxide minerals—e.g., silica, SiO2, as in quartz.
(Interestingly, these 4 cations all serve as essential vertebrate
micronutrients.) However, the crustal abundance of Si4+ far
exceeds that of all the others in this group. Thus Si4+ appears in
large quantity in both the insoluble products of weathering, most notably as
sand, and in natural waters as dissolved silica. Si4+ binds to igneous
minerals only at intermediate to low temperatures and remains abundant in fluid
phases to the end of the crystallization sequence. Along with their low
densities, these properties insure the crustal accumulation of quartz and silicates during the earth's chemical
differentiation.
Hard vs. Soft Cations
Another important innovation in Railsback's revamped
periodic table is the grouping of ions according to their electronic
configurations as ions, a dimension separate from ionic potential. Having
lost all their outer shell electrons, the hard cations are left with a
relatively inert noble-gas-like electronic configuration, while the soft
cations retain some outer shell electrons—the more, the softer. Hard and soft
cations behave quite differently. Hard cations like Ca2+ coordinate
strongly with O-2 and F-; soft cations do not. When they
form oxide minerals, hard and soft cation oxides have high and low melting
points, respectively. Soft cations like Cu+ bond with S-2
and the larger halides I- and Br- rather than with O-2
and F-; they tend not to form oxide minerals. Thus hard Ca2+
forms both oxide and an oxygen-rich sulfate (gypsum, CaSO4) but not a
sulfide, while soft Cu+ forms a sulfide (chalcocite, Cu2S)
but not an oxide.
The metal cations
commonly found in silicate minerals (Na+, K+, Ca2+,
Mg2+) are all hard. Their low ionic
potentials and noble-gas-like electronic configurations allow them to fit
cleanly between large polymeric silicate and aluminosilicate sheets and chains and bind
them together
without disturbing them. The trace elements, on
the other hand, generally have low to intermediate ionic potentials and soft to
very soft outer shell configurations. Due primarily to the latter, they fit
poorly in silicate lattices and for the most part remain
sequestered in the mantle, where more hospitable non-silicate minerals dominate.
Anions
The most commonly occurring anions
(negatively charged ions) are O-2, S-2, Cl-, F-,
and the soluble oxo complex radicals CO3-2, SO4-2,
SiO4-4, NO3- and PO4-3.
Hard cations prefer to coordinate with O-2, by far the most common
anion in both crust and mantle, while soft cations like
Pb2+, Cu2+ , Zn2+ and Ag+ instead prefer S-2
(number 13 of the 14 most common elements. This preference alone accounts for
some of their rarity. The extremely soft Au+ cation can't form an oxide and can only form a
sulfide with the help of other soft cations—hence the long-admired rarity and "nobility" of gold and its predilection to
go native in elemental form.
Not surprisingly, metal oxides and sulfides are the most common ore
minerals in the Colorado Mineral Belt and
elsewhere. After O-2 and S-2, the properties of the anions
appear to be less important than those of the cations.
Basic Rock Types
Now that we've seen how crustal
elements combine to form minerals, let's look at the rocks the minerals
make.
Every grade-schooler knows that rocks come in
three basic flavors—igneous, sedimentary
and metamorphic, as detailed in the table
below. That's still an excellent starting point, but we'll need some
subtypes to make real headway in understanding the rocks of Colorado. It's worth
emphasizing at the outset, however, that rocks in the field form a
continuum of origins, compositions and textures beyond the reach of any rigid
classification scheme. No matter how fancy the classification, there will always be important transitional rocks that
can and will be classified more than one way by reasonable geologists.
Basic Rock Types
|
| Type |
Defining Characteristics |
Common Features |
Subdivisions and Examples |
Common Features |
|
Igneous
|
Solidified from a molten or partially molten
state
|
- Interlocking grains
- Lack of fossils
- Chilled margins against adjacent rocks
- Thermal alteration of adjacent rocks
|
Extrusive
(volcanic)
|
- Respect for pre-existing surfaces
- Fine-grained
- Layering
- Vesicles
|
| Intrusive
(plutonic)
|
- Cross-cutting of pre-existing structures
- Coarse-grained
- Lack of layering
- Lack of vesicles
|
|
Sedimentary
|
Consolidated
from loose materials or solutes deposited by ice, water or air, including
the remains and secretions of living organisms
|
- Stratification
- Sorting
- Fossils
|
Clastic
|
- Chemically heterogeneous
- Internal structures reflect depositional environment
|
| Chemical
|
- Chemically homogeneous
- Little internal structure
|
|
Metamorphic
|
Derived from pre-existing rocks altered by heat,
pressure or exposure to fluids, usually in the solid state
|
- Distorted internal structures such as fossils
- Parallelism among elongated grains
|
|
|
Transitional Rock Types
Much to the dismay of architects, students and users of
rock classifications, transitional rock types pop up everywhere. Important examples include
the following:
-
Mildly
altered sedimentary rocks may still look just like sedimentary rocks, but some
geologists will label them metamorphic where others would not.
-
Pelagic cherts derived from
planktonic debris could just as easily be considered biochemical
rocks, but by convention, they're classed as chemical
because of a necessary recrystallization step.
-
Marls are mixtures of clays
and carbonates with at most minor amounts of quartz.
Are they clastic or chemical
sedimentary rocks?
-
Tuff—volcanic ash
deposited in layers as it falls out of the air onto land or water—could be
considered sedimentary, but by convention, tuffs are classed as
igneous.
-
Volcaniclastics
formed from debris weathered from solid volcanic
rocks have some unique properties. They might be considered igneous,
but they're usually classified and mapped as clastic sedimentary
rocks.
-
Migmatites
contain some melted rock but are still considered metamorphic, not
igneous.
-
Cumulates—masses of
precipitated crystals settling to the bottom of an otherwise fluid magma—are
igneous sediments in a very real sense, but rocks formed exclusively from
cumulates are considered igneous, not chemical
sedimentary.
Confused? Hang around with rocks long enough, and you'll get used to it.
Crystalline Rocks
Rocks composed entirely of interlocking crystals of
one or more minerals are said to be crystalline. All unequivocally
igneous rocks are crystalline, as are higher-grade, recrystallized metamorphic
rocks like gneiss and schist.
Strictly speaking, some chemical sediments like limestone and
chert
(microcrystalline quartz) are also composed of crystals, but in common usage,
the term excludes sedimentary
rocks.
Mechanically, crystalline rocks tend to be stronger and more
resistant than other types. They form the crests of the Rockies' highest ranges
and hold up most of Colorado's Fourteener summits.
Rock Stability and the Rock
Cycle
For the most part, rocks are equilibrium
products relatively stable at
the conditions under which they formed but chemically or mechanically unstable
in all other environments. Crystalline rocks formed
at depth are unstable at the surface, while sedimentary
rocks formed at the surface are unstable at depth.
Rocks thrust into new settings by tectonic,
magmatic or erosional events tend to move around the rock cycle
(below) from one
basic rock type to another. For example, igneous rocks formed at depth
under high temperature (T) and pressure (P) in the absence of free oxygen and water
are bound to change when brought to the surface to face chemical and mechanical weathering,
erosion, transport, deposition
and diagenesis. Given enough time, their debris will become sedimentary rocks
best suited to
surface conditions. With deep burial during a mountain-building event, the elements
in the sedimentary rocks will reorganize into new metamorphic
minerals better suited to the extreme pressure-temperature (PT)
conditions they now face. Under the
right PT conditions, they might even come full circle to melt back into igneous
rocks. Alternatively, uplift and erosion of the metamorphic
rocks might ultimately produce a new batch of sedimentary rocks. All paths through the
rock cycle are possible.
Rock Cycle
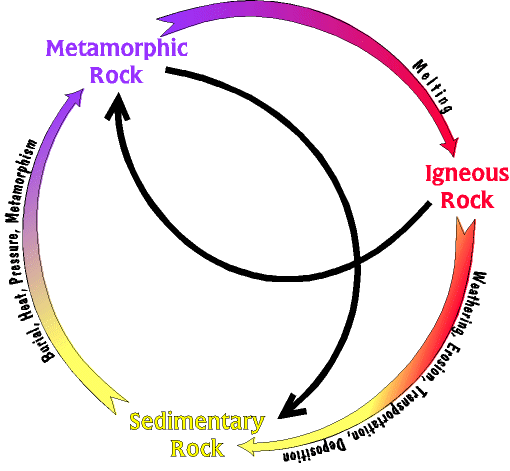 |
Rock Cycle
|
The
diagram at right nicely summarizes the rock adjustments outlined above. It's
useful to think of igneous rock as the starting point for the
cycle, but material can jump in anywhere and end up anywhere. The transformations that occur with
any frequency are already shown in the diagram, but since
deeply buried sedimentary rocks can melt directly in certain tectonic environments, it would be reasonable to add another thin
black curved arrow pointing from the sedimentary to the igneous node.
Acknowledgment: Rock cycle diagram courtesy ^Lynn
Fichter.
Resistance to Weathering
Rocks that are slow to weather and
erode are said to be resistant.
All other things being equal, erosion will leave resistant rocks
standing higher than the less resistant rocks around them. Crystalline
(igneous and metamorphic) rocks then to be more resistant than unaltered
sedimentary rocks, but chert is a notable exception.
Fusibles
and Refractories
Fusibles are rocks or minerals that melt easily.
Refractories are just the opposite. Sedimentary rocks tend to be
fusible, while crystalline rocks tend to be refractory, some more than others.
Not surprisingly, the most refractory rocks, like gabbro and peridotite,
reside in the lower crust and mantle.
Sedimentary rocks groomed for surface stability
wouldn't stand a chance at those depths.
Reworking
Rock materials don't necessarily move around the
rock cycle as the rocks they compose evolve. In a tectonic disturbance, uplifted
sedimentary rocks can be reworked into new sediments, igneous rocks can remelt,
and metamorphic rocks can prograde. Reworking adds yet another layer of
complexity to rock genealogy.
End-Stage Products of Weathering
Many earth processes play out at depth
beyond the reach of the atmosphere and hydrosphere, but for many others (including
weathering, erosion, transport,
deposition, isostatic rebound
and basin subsidence), the rubber meets the road at the surface, where the atmosphere,
the hydrosphere and the land all interact strongly to shape both land and climate in a
never-ending dance.
Once weathering gets a foothold on a rock exposure,
erosion, transport and deposition
are likely to follow, but
weathering continues to break down the sediments, both en route and at the site
of deposition. Given sufficient
time and transport distance, the ultimate end-products of weathering are always pretty
much the same, regardless
of the initial rock type:
-
Quartz (SiO2) sand,
typically derived from igneous, metamorphic and reworked quartz-bearing
sedimentary rocks like sandstones and quartzites
-
Clay mud, from the chemical breakdown of
feldspars and ferromagnesian minerals in igneous, metamorphic and immature
sedimentary rocks, and from reworked clay-bearing sedimentary rocks
-
Dissolved calcite (calcium carbonate, CaCO3,
AKA lime), derived from calcium (Ca) weathered from common feldspars,
and from reworked lime-bearing sedimentary rocks
Why end up with just these three mineral groups? Because all the rock-forming
minerals commonly exposed on this silicate planet eventually break down into quartz, clay
or calcite unless some other process (like melting) intervenes. These
end-products are chemically stable under most surface and near-surface
conditions, but their precursors are not.
Quartz Grains
Quartz (SiO2) grains are exceptionally stable at the surface. They
may be ground down to silt-size during transport, but like glass (also SiO2),
they're chemically inert. (That's why chemists use glass containers.) Once
silt-sized, they go back into suspension in moving water, where they escape further
mechanical weathering.
Clay Minerals
Clay minerals tend to form microscopic flat platy crystals with charged surfaces
that slide easily against each other and have a hard time interlocking,
especially when wet. Claystones tend to be weak as a result, and clay particles
remain in transport the longest because of their size and shape. The most common clay minerals produced
by weathering—montmorillonite, illite, kaolinite, in
order of current abundance—reflect the stability of sheeted Si-O and Al-OH
crystal structures. Montmorillonite is the expansile clay dreaded by homeowners
and civil engineers everywhere. Kaolinite formation is restricted to low latitudes because it
requires a hot wet climate.
Map Units
Some sections in this article close with a "Map Units" subsection
describing how to find pertinent bedrock (surface rock) units on the Geologic Highway Map of Colorado.
Top
Page Index
Igneous Rocks
Rocks that solidify from a molten or partially molten state are said to be igneous.
Rocks that freeze on or above the surface, whether in the air or underwater, are extrusive
or volcanic. But if they freeze below the surface for any reason, as did
the 1.4 Ga Silver Plume granites exposed so handsomely at ^Rocky Mountain National
Park (right), they're
called intrusive
or plutonic instead. As we'll see, extrusive and intrusive igneous rocks
differ chemically, texturally, and in other important ways. In either case, magma is the molten rock involved. Magmas
reaching the surface in liquid state are called lavas.
For more on igneous rocks, read on, but also
consider a visit to the extensive and
well-illustrated ^Igneous
Rocks site by educator and geologist ^Lynn
Fichter.
Melts — Buoyant and Reactive
The geothermal gradient guarantees
that all melts develop at depth. Since melts are almost always lighter than the
solid rocks from which they derive, gravity impels them to rise toward the surface as best they
can, just as a bubble eventually rises, however slowly, through semi-solid
molasses in the refrig. (A rising rock body, whether solid or molten, is a diapir.)
In fact, most rock melts are buoyant enough to approach the surface if they
don't freeze first.
On the way up, melts interact both physically and chemically with the rocks
through which they pass. In the process, they give off heat and fluids and take
in easily melted or dissolved wall rock components. The final igneous product, whether
extrusive or intrusive, is
always highly evolved relative to the initial melt, but some magmas
reaching shallow levels are more primitive than others. On average, the basalts
erupted at seafloor spreading centers are the least evolved relative to their asthenospheric
source rocks. Continental granites and rhyolites
are among the most differentiated of all magmas.
Igneous Settings
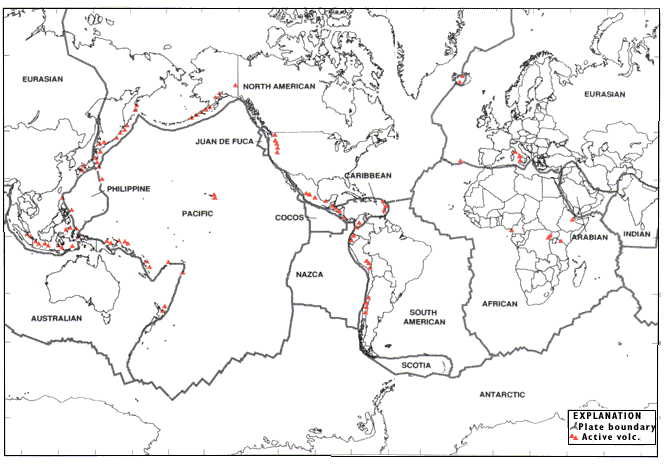
Since the onset of plate tectonics ~2.0
Ga, most of the planet's igneous activity has concentrated along plate
boundaries. As the modern map below clearly shows, the situation is no different
today. Unusually eruptive boundary segments like Iceland
are called hot spots. The igneous
centers found far from plate boundaries are also hot spots (Hawaii
is the only one shown here, but others exist.) Hot spots arise for a variety of
reasons, most of which ultimately relate to extensional failure of the plate(s)
involved; the once-popular deep mantle plume
explanation for hot spots is not supported by the observations. Volcanic outputs
at hot spots can be truly prodigious, but the associated intrusive
activity can be just as important.
Raw Materials For the Rock Cycle
In many ways, igneous rocks are the starting point for the rock
cycle. Whether the parent melt derives from the mantle or from
sedimentary rocks buried deep in the upper
crust, it eventually cools and freezes into a solid mass of interlocking crystals
derived from a handful of mineral families, including those listed in the table
below. The igneous raw materials can then go on to become sedimentary
or metamorphic rocks as events and conditions unfold.
Mineral Families Commonly Found in Igneous Rocks
|
| Composition |
Color |
Mineral Family |
Common Members |
| Felsic |
Light |
Quartz
|
Quartz
|
|
Feldspars
|
Plagioclase, orthoclase, microcline
|
| Feldspathoids* |
Leucite, nepheline, sodalite, cancrinite,
melilite |
| Mafic |
Usually Dark |
Ferromagnesian minerals
|
Olivine, pyroxene, hornblende, amphibolite
|
|
Micas
|
Biotite (black), muskovite (white), phlogopite (black)
|
* Table Note: Since quartz and feldspathoids (AKA foids) are chemically incompatible,
they seldom occur in the same rock. Most of the remaining possible mineral
family combinations can and do occur in common rocks.
Felsic and Mafic Rocks
The term felsic means feldspar- and silica-rich. Sialic rocks
(those rich in silica and aluminum) are particularly felsic. Felsic rocks tend
to be of continental origin. Felsic magmas like rhyolite have typically reacted
strongly with continental (or at least felsic) crust on their way to the
surface, regardless of the source of melt.
Mafic means Mg- and Fe-rich.
Rocks of the upper continental crust are
felsic on average, but mafics are fairly common there. Rocks of the lower
continental crust and the oceanic
crust are almost always mafic. Mafic rocks are on average denser than
felsics and tend to be found at greater depths as a result. Ultramafic
rocks are exceptionally rich in Mg and Fe and poor in Si. They almost always
come from the mantle and accordingly tend to be very dense.
Magmatic Differentiation
A single melt can produce a wide variety of different igneous rocks through a
complex process known as magmatic differentiation. Exposed magma bodies, whether
intrusive or extrusive, typically display some degree of differentiation over
both space and time. Most melts develop in the lower
crust or in the upper mantle's asthenosphere.
As a result, many melts start out with a fairly primitive mafic or basaltic
composition. Melts developing in the upper crust tend to have higher initial
silica contents
Regardless of where they form, all melts evolve considerably during ascent.
The rocks they ultimately produce depend on the composition of the original melt
and on the properties of the wall rocks encountered en route. The main processes
involved are fractional crystallization, assimiliation,
exchange of volatiles, and magmatic
mixing.
Fractional Crystallization
Fractional crystallization (or fractionation for short) occurs when
circumstances prevent early-forming crystals from
reacting with the remaining melt. This process accounts for most of the differentiation
observed in igneous rocks.
As a rising melt cools and reacts with surrounding rock, the melt minerals
with the highest melting points or the lowest solubilities (the refractories,
like olivine and pyroxene) crystallize out first, while those with the lowest
melting points or solubilities (the fusibles, like silica) freeze out last. Heat released by
the crystallization of refractories replaces heat lost to the surrounding country
rocks by simple conduction, by country rock melting and by the assimilation of
country rock fusibles. Fusibles
and refractories enter and leave the melt at specific temperatures and
pressures, which tend to occur at specific depths along the ascent. As it
continues to rise, the surviving melt
loses volume, and its fusibles become more and more concentrated. It leaves
behind a trail of solid refractories and country rock alterations.
Gravitative differentiation, the most common
form of fractionation, stems from the fact that most solid minerals are
more dense than their parent melts. When their crystals settle to the bottom of the
magma body, they are effectively segregated from the residual melt. Rocks formed
from crystals amassed in this manner are called cumulates, and they're
often zoned, with first crystals to leave the melt at the very bottom of the
magma chamber. Cumulates formed from lighter crystals that occasionally
precipitate out of the melt float to the top instead, with the lightest at the
very top. Cumulate crystals are typically cemented by residual magmatic
fluids.
Assimilation
Ascending
magmas also evolve chemically by recruiting easily melted or dissolved
components (fusibles) from the walls of their conduits. Heat and magmatic fluids
mediate the process. In so doing, they may
pick up volatiles, extra silica, trace
elements and even chunks of wall rock. The
thermodynamics and geochemistry involved are exceedingly complex, but the heat
the melt gains from leaving behind refractories (an exothermic process) is
usually sufficient to cover the heat lost to the endothermic reactions involved
in the assimilation of country rock components. Assimilation can thus proceed
without tapping the heat required to keep the melt from freezing.
 |
Partially-assimilated xenolith in granite boulder,
Glenwood Canyon
|
Wall rock chunks that
survive more or less intact, without completely melting or dissolving into the
magma, are called xenoliths. Surviving wall rock crystals are called xenocrysts.
Together, xenoliths and xenocrysts provide invaluable information about the
rocks residing at rarely exposed lower crust and mantle levels.
Volatiles, Aplite and Pegmatites
Figuring prominently in the process of assimiliation are the volatiles found in varying amounts in nearly
all wall rocks and magmas—CO2, SO2, O2,
Cl2 and most notably, H2O. Water is particularly available
in wall rocks of the mid-crust, both in free form and within the hydrated minerals
commonly found at such depths. Some of the assimilated water goes
into hydration reactions with predominantly anhydrous melt components, but most
of it just builds up in the ever-shrinking surviving silicate melt. If it takes
on enough water, the melt will eventually develop a water-saturated
silicate fraction and a separate water-based fluid phase.
 |
Pegmatite vein in granite boulder, Glenwood Canyon
|
Under certain conditions, the
water-saturated silicate fraction can give off a whitish fine-grained
vein-filling slurry of quartz and feldspar known as aplite. The
water-based phase easily assimilates trace elements that don't fit well in
most silicate crystals, including
lithium, beryllium, niobium, tantalum, tin, uranium, thorium, tungsten,
zirconium and the rare earths. Many ore deposits form when this hot,
pressurized, mineral-laden hydrous fluid finally permeates fractured country
rock and cools into veins of pegmatite—an igneous rock containing
unusually large crystals of quartz, feldspar and, now and then, highly prized
minerals as well. Pegmatite and aplite dikes and veins are common around intrusions.
Pegmatite is the prospector's friend.
Magmatic Mixing
[photo coming]
|
Banded tuff, Valley of 10,000 Smokes
|
The mixing of two separate magmas just before eruption or final subsurface
emplacement is uncommon, but in areas of active magmatism, adjacent magma bodies
are bound to develop transient subsurface communications now and then. At right is a rare banded tuff from the Valley of 10,000 Smokes, Katmai,
Aleutian Archipelago, Alaska. The banding reflects the last-minute mixing of
lavas from two separately differentiated magma chambers underlying the valley during the cataclysmic
1912 eruption of Novarupta Volcano,
which released a whopping 30 km3 of pyroclastic material at the time.
[banded tuff photo]
A much more common form of magmatic mixing involves the secondary melting (anatexis)
of mid-
to lower crustal rocks on contact with much hotter rising mafic melts of mantle
origin to produce felsic (feldspar- and quartz-rich) magmas in arc
and continental rift settings. On reaching high crustal levels,
such melts may arrive with more mantle
heat than mantle material in tow.
Eruption
When magma reaches the surface, the excess
volatiles escape in
vapor form. Gases usually boil out of low-viscosity basaltic
lavas relatively peacefully, as they usually do in Hawaiian eruptions. A good
example shown at right is the ^March
26, 1984 fissure eruption on Mauna Loa's Northeast Rift Zone. But volatiles are more
likely to explode than boil out of viscous lavas like rhyolite
and andesite,
as they did at Mt. St. Helens on May 18, 1980 (shown at the top of the next
section). Volcanic habits are discussed in greater
detail below.
Top
Page Index
Volcanic (Extrusive) Igneous Rocks
Igneous rocks
that solidify from melt on or above the surface of the solid earth are called volcanic
after Vulcan, god of fire. The term extrusive is synonymous with
volcanic. At right is the explosive June 12, 1991 eruption of Mount Pinatubo,
Luzon, Philippines.
Volcanic rocks occur in many tectonic settings, including magmatic
arcs at subduction zones (as in the Banda
Sea at right), seafloor spreading centers,
ocean islands, and along
continental rifts and other leaky faults. Like their intrusive
counterparts, extrusive rocks are categorized primarily on the basis of
texture and composition.
Volcanic Textures
 |
Pahoehoe lava, Mauna Ulu, Hawaii
|
Because volcanic rocks tend to cool quickly after eruption, individual
mineral crystals have little time to grow and usually end up too small to see with the
unaided eye. The resulting rock texture is said to be aphanitic. Occasionally,
one mineral, often a feldspar, manages to grow phenocrysts (large
crystals much bigger than all the rest) before venting. An
otherwise aphanitic volcanic rock containing phenocrysts is called a porphyry;
the fine-grained component is called the groundmass. Rock textures in which two very
different grain sizes predominate are termed porphyritic. Volcanic
glasses like obsidian tell of ultra-fast cooling rates.
Lavas
 |
Sunrise, Mauna Loa, Hawaii
|
Lava flows are perhaps the
simplest of volcanic deposits, but they show their share of complexities.
Between the frozen gas bubbles (vesicles), if any, most lavas are
predominantly aphanitic in texture, but porphyries also occur. Over time, flows
tend to vary in texture and composition, in part because they tap different
portions of the magma chambers that feed them. Flows often cross eroded surfaces
and interact with their soil covers and groundwater along the way. Lavas
quickly chilled in air or water develop glassy textures. ^Obsidian
(usually of rhyolitic composition) and the glassy
rinds on basaltic pillow lavas are examples.
At right, Mauna Loa looms over Kilauea Caldera at ^Hawaii
Volcanoes National Park as fume rolls off Steaming Bluff in the morning
light. Mauna Loa is the world's largest mountain and largest volcano.
Kilauea is the world's most active volcano. Basaltic
lavas built both just in the last 1 Ma. Olivine basalt featuring macroscopic
green olivine porphyrocrysts in a black groundmass is a common lava around Mauna
Loa. Waves and currents have concentrated olivine dense crystals weathered out
of sea cliffs at the southern tip of the Big Island into a unique green sand
beach.
Tephra
[photo coming]
|
Banded tuff, Valley of 10,000 Smokes
|
Solids
thrown from a volcanic vent are called ejecta, and accumulations of ejecta are
called tephra
or pyroclastic deposits. Pyroclastics come in many sizes: Blocks
and bombs are over 32 mm in diameter, with bombs showing some degree of
aerodynamic rounding; lapilli are 4-32 mm across;
and ash particles are under 4 mm. Wind can carry fine ash hundreds of
kilometers, but, not surprisingly, larger and larger ejecta fall progressively closer to the vent. Tuff, rock made
from consolidated ash layers, comes in water-laid and air-fall
varieties. When ash is hot enough and falls at high enough rates,
individual particles can fuse on burial by subsequent ash falls. An extremely resistant welded
tuff or ignimbrite results.
Around 30 Ma, welded tuffs blanketed the
entire Basin and Range to great thicknesses in a prolonged and undoubtedly
unpleasant event known as the Ignimbrite
Flare-up. During the Early Phase of
Tertiary magmatism in Colorado (~37 Ma), massive ash flows from the Mount
Princeton area rolled 90 km across the Eocene
erosion surface to blanket the western Denver
Basin with incandescent ash which compacted and fused to become the welded Wall
Mountain tuff, apparently in a single day. Given the 10:1 compaction ratios
typical of welded tuffs, modern Wall Mountain remnants up to 40' thick imply
initial ash deposits up to 400' thick.
[photo coming]
|
Wall Mountain ignimbrite clast from Castlewood Canyon near Franktown,
CO
|
I keep the clast of Wall Mountain tuff pictured at right on my desk to help
me remember what a really bad day looks like.
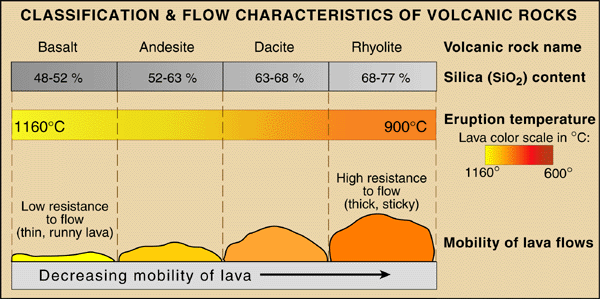
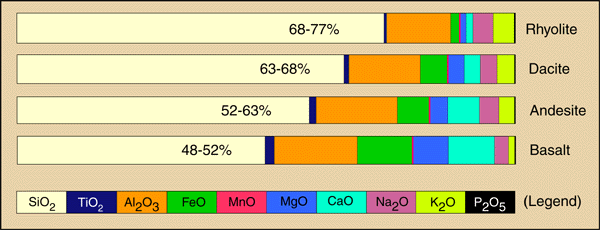
Volcanic Compositions
Volcanic rocks vary widely in their elemental and mineral content. Silica
content is perhaps the important single compositional property because it
strongly controls viscosity. Viscous high-silica
lavas like rhyolite
and andesite
tend to erupt explosively, because stickier lavas retain more of their volatiles
until they near the vent and release them more violently on eruption. As with
crustal rocks in general, silica (SiO2) and alumina (Al2O3)
dominate all volcanic rocks, even the most mafic basalts,
but the silica variations shown below are more than adequate to create big
differences in lava viscosity.
 |
|
|
In order of increasing silica content and
explosive tendency, basalt, andesite, dacite
and rhyolite
are the most common lavas. As the lava of choice of at mid-ocean
ridges, on seamounts and submarine
plateaus, along continental rifts and
other leaky faults, basalts outnumber and outmass all other lava types by a wide margin.
Basalts are mafic (rich in ferromagnesian minerals), whereas rhyolites
are felsic (rich in high-silica feldspars, feldspathoids and quartz).
Andesites and dacites are in between. Compositionally, rhyolite is the extrusive
form of granite, while basalt resembles the intrusive
rock gabbro.
Common Lavas
|
| Lava |
Silica Content (by weight) |
Viscosity and Tendency to Explode |
Common Minerals |
Eruptions temperatures (°C) |
^Basalt
|
48-52%
|
Low |
Olivine, pyroxene and plagioclase
|
1,100-1,250
|
^Andesite
|
52-63%
|
Medium |
Plagioclase, pyroxene, lesser amounts of
hornblende, sometimes olivine
|
900-1,100
|
| ^Dacite |
63-68% |
High |
Plagioclase, pyroxene, and amphibole |
800-1,000 |
^Rhyolite
|
68-77%
|
Very High |
Quartz, feldspar and biotite
|
700-850
|
Table note: The external links above lead to excellent rock photographs and descriptions
provided by the US
Geological Survey Photo Glossary of Volcano Terms.
For the visual learner, the diagram below shows much the same information.
Volcanic Habits
The way a particular volcano behaves depends on many things, including
-
the composition, temperature and viscosity of the lava
currently being erupted
-
the vigor of the magma supply
-
the load of dissolved volatiles
(gases) to be released
-
the availability of free water (surface or
ground,
liquid or solid) to enhance the potential for explosive eruption and the
generation of landslides and mudslides.
Of these, the most important influences relate to the properties of the lava
itself.
Basalitic Volcanism
 |
Mauna Loa, 1984
|
Immature oceanic arcs usually start
out with basaltic lavas. Seafloor spreading
centers, ocean island volcanoes, volcanic
seamounts and volcanic submarine plateaus
overwhelmingly produce basalts. Basalt is also the magma of choice in continental
rift settings, at least for starters, and along leaky faults of all kinds.
Subaerial basaltic volcanoes generally have limited potential for devastation
at a distance. Their lavas tend to erupt at high temperatures with low
viscosities, the latter due to their low silica contents, and their products
tend to accumulate near the vent. That makes for fairly peaceful eruptions and
for relatively stable volcanic edifices, including shield volcanoes like Hawaii's
Mauna Loa, shown at right in a 1984 fissure eruption. Note the calm observer
in the lower left corner of the photo. Standing that close to an erupting vent
would be totally insane on a non-basaltic volcano.
Still, even basaltic volcanoes have their moments. Groundwater meeting hot rock
beneath the Kilauea Caldera caused a massive week-long phreatic
(steam-driven) eruption that blasted Halemaumau
crater out of the caldera floor in 1924. When the dust had settled, the new
crater was 915 m across and 550 m deep, and 750,000 m3 of ejected
debris littered the caldera floor, including many angular car-sized blocks.
Arc Volcanism
 |
Mt. Rainier looms over Takoma, Washington
|
Andesites, dacites
and rhyolites are the magmas most commonly erupted at magmatic
arcs associated with subduction zones,
particularly those built on continental margins. These viscous lavas tend to
build unstable stratovolcanoes—like Mt. Rainier (right) of the Southern Cascade
Range—prone to release massive lahars (volcanic mudflows) and
ground-searing ashflow (glowing cloud) eruptions. As Mount St. Helens so
vividly reminded us, they also tend to erupt explosively, potentially spreading
large volumes of ash over wide areas. The apocalyptic "X"
bentonite layer of the Denver Basin
records the fall of 15 m (48') of ash from an Idaho vent hundreds of kilometers
upwind.
Bimodalism
Most arcs and many rifts go through the full range of magmas—first
basalt, then andesite, then dacite
and finally rhyolite—as source melts interact with wall
rocks on the way up to their reservoirs, and as the magma
reservoirs mature over time. But volcanic centers in both rift and arc
settings not uncommonly jump straight from basaltic to rhyolitic products. If
intermediate magmas are largely absent, the eruptive sequence
is called bimodal. Bimodal sequences used to be taken as markers for
continental rifting, but many bimodal arc sequences have been recognized as
well.
Volcaniclastic
and Metavolcanic Rocks
Clastic sediments derived from solid volcanic source rocks
are called volcaniclastic and are usually classified and mapped as sedimentary
rather than igneous. Volcaniclastics and their metamorphic
derivatives deserve special mention here as an important transitional
rock type in Colorado's physical evolution.
Clastic volcanic sediments can accumulate in large volumes
around active magmatic arcs and volcanic
fields. Their fine-grained feldspar and ferromagnesian minerals quickly weather
to clays, and their free quartz crystals are already silt-size or
smaller. Arkose (feldspar-rich) and lithic
sandstones are common in immature volcaniclastic sediments deposited near the
source, while silty mudstones are common among mature volcaniclastic sediments.
Immature volcaniclastics shed to the east
from early Laramide volcanic
highlands in the Front Range uplift accumulated to form the Denver, Arapahoe
and Dawson Arkose formations at the top of
the Denver Basin.
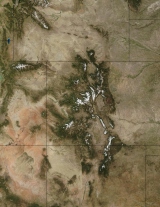
Metavolcanics
 |
1.7 Ga metavolcanics
|
Under regional metamorphism,
volcaniclastics tend to evolve along the shale
pathway detailed below. In metamorphic form, arc-derived
1.7 Ga volcaniclastics make up a good share of Colorado's
Precambrian basement, including the craggy hornblende gneiss exposed atop Royal
Mountain (right).
Volcanic Map Units
On the Geologic Highway Map of Colorado, the
surviving volcanic outcrops are all Tertiary in age. The Oligocene andesites
labeled "Tov" in brown are by far the most extensive and occur
primarily in the San Juan and Thirtynine Mile volcanic fields. Widespread
Miocene-Pliocene basalts and bimodal products are mapped as "Tuv" in
orange. The legend shows a Quarternary basalt unit "Qv" in stippled
orange, but I have yet to spot a mapped occurrence—not even at the Dotsero
Volcano, last active about 4 Ka. (Tweeto's 1979, 1:500,000 Geologic Map of
Colorado correctly shows a Quaternary basalt unit there.)
Top
Page Index
Intrusive (Plutonic) Rocks
By
definition, intrusive
igneous rocks solidify from a melt before reaching the surface. The term plutonic
(after Pluto, god of the underworld) is synonymous with intrusive. Coherent
bodies of plutonic rock are called intrusions. The host rocks surrounding
the intrusion are referred to as country rocks. Intrusives are often involved in the
plumbing system of a volcanic edifice or field, as
were the beautifully exposed Oligocene intrusions seen at right in the West
Elk Mountains, but some never communicate with the surface while still
molten. Like their extrusive counterparts, intrusive
rocks are categorized on the basis of
texture and composition.
The
highest point on the horizon in the photo at right is Mount
Evans (14,264') as seen from Denver. Its summit area exposes a large
intrusion (batholith) of resistant granite
emplaced during the 1.4 Ga Berthoud Orogeny,
perhaps the most common intrusive rock type found
in Colorado.
Intrusive Textures
Slower subsurface
cooling times lead to larger average intrusive grain sizes—much larger than
the microscopic grains typical of extrusive (volcanic) rocks. Magmas solidifying underground tend to cool very
slowly, in part because the already warm solid
country rocks surrounding the intruding magma are effective thermal insulators.
Individual
crystals commonly reach 2-4 mm diameters, but in pegmatites,
the
quartz and feldspar crystals can exceed 100 mm. Such igneous textures are called
phaneritic.
The grains in the granodiorite bolder at right are
easily visible with the unaided eye. This particular boulder came from an
Oligocene intrusion exposed along the lower Cathedral Peak in the Elk
Range.
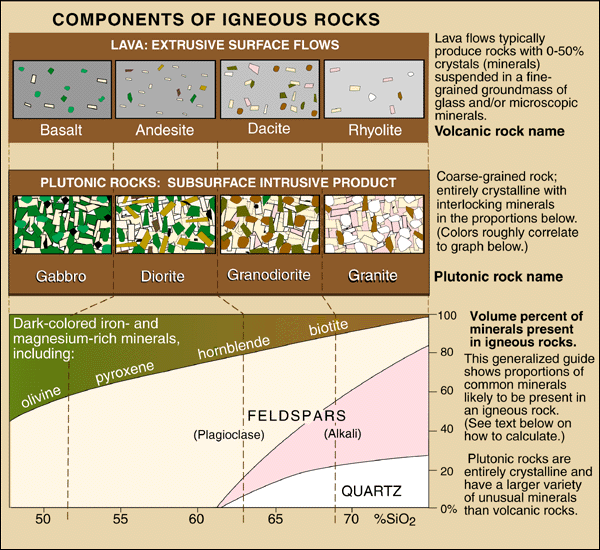
Intrusive Compositions
Most intrusive rocks contain the minerals shown in the diagram below. From
left to right in the mineral plot at the bottom of the diagram, the rocks go
from mafic (Mg- and Fe-rich) to felsic (feldspar- and silica-rich)
in composition.
A mix of light and dark grains is typical in intrusive rocks. The
light-colored grains include quartz (usually colorless to gray) and one or more
feldspars (off-white to pink, green or gray). Micas, if present, tend to be either black
(biotite) or white (muscovite). Olivine, pyroxene and hornblende range from
green to black. In intrusive rocks, all these minerals have characteristic
crystal shapes readily observable with a hand lens when present, but the
individual crystals aren't always well developed.
Common Intrusive Rocks
Intrusive rocks are easily recognized as such but difficult to classify,
particularly in the field. The classification schemes used by geologists are far
too
complex to discuss there, but the most common intrusive rocks are worth
exploring.
Most of the intrusives found in Colorado are light-colored felsic
(feldspar- and silica-rich) to intermediate rocks along granite-granodiorite
lines. Many of the Colorado rocks called and even mapped as "granites"
turn out to be granodiorites or other felsic types on closer inspection. A few mafic
(Mg- and Fe-rich) Laramide
intrusives have been found in Colorado, but mafic intrusions are not
uncommon worldwide.
Common Igneous Intrusive (Plutonic)
Rocks
|
| Rock |
Composition |
Notes |
Notable Colorado Occurrences |
| ^Granite |
- Quartz > 20%
- Feldspar = orthoclase + plagioclase
- Mica = biotite ± muskovite
- Occ. hornblende
|
- The quintessential light-colored quart-dominated felsic continental
rock
- If plagioclase > orthoclase, it's called tonolite or plagiogranite
- If orthoclase > plagioclase, it's an alkali granite
|
|
| ^Quartz
monzonite |
- Quartz 5-20%
- Feldspar = Na plagioclase ± orthoclase
- Mafics < 10%
|
- Light-colored due to low mafic content
- A feldspar-dominated felsic rock; may contain nothing but feldspar
- If quartz < 5%, it's just a monzonite
- Anorthosite, a rock that hasn't formed since the early
Precambrian, is ~100% Ca plagioclase
|
|
| ^Granodiorite

|
- Quartz > 20%
- Feldspar = Na/Ca plagioclase
- Biotite + amphibole = 10-25%
|
- An intermediate rock with high quartz and mafic content, an
unusual combination
- With quartz < 5%, it would be a diorite
instead
- Salt-and-pepper appearance
|
|
| ^Diorite |
- Quartz ~ 0%
- Feldspar = Na/Ca plagioclase
- Amphibole ~ 50%
- Occ. biotite
|
- An intermediate to mafic rock
- If quartz reaches 5-20%, it becomes granodiorite
- Orthoclase is absent
- Mafic content is too high to be a monzonite
- Salt-and-pepper appearance
|
|
| ^Gabbro |
- Feldspar = Ca plagioclase
- Pyroxene ~ 50%
- Freq. amphibole
- Occ. olivine
|
- The most mafic of the common intrusives
- Dominates the lower continental
and oceanic crust
- Usually dark-colored
- Diabase, a finer-grained version of gabbro, is a common fill
in dark dikes
|
|
Table Note: The external links above lead to Lynn Fichter's
well-illustration igneous rock descriptions.
Common Intrusive Forms
Depending on the composition of the magma, the final depth and the nature of the country rocks,
intrusions can take many forms.
Common Intrusive Forms in Colorado
|
| Form |
Definition |
Notes |
Notable Colorado Occurrences |
|
Diapir
|
Any rising rock mass; if molten, the diapir could give rise to any of the
intrusive forms listed below
|
[] |
[] |
|
Diatreme

|
A volcanic pipe or neck blown out through overlying country rock by a gas-driven
subsurface explosion and filled with country rock breccia; in some cases, the
gas may be steam flashed from ground water (a phreatic eruption)
|
Diatremes can be partly extrusive in the sense
that their breccias may include erupted materials that fell back into the
conduit. Eruptions of kimberlite, a rare CO2-charged,
diamond-bearing magma originating in the upper mantle, commonly form
diatremes |
- ^Ship
Rock (shown)
- Cripple Creek gold field
- State Line diatreme field
|
| Stock

|
A cylindrical intrusion with no known floor
and a surface exposure under 100 km2 |
Stocks may serve as major feeders for large volcanic
fields |
|
|
Pluton
|
A moderate to large frozen intrusive body, usually in the shape of an oblate spheroid,
which may or may not have been associated with volcanism
|
In Colorado, plutons seem to be less common
than the larger and smaller intrusive bodies listed here. |
|
Batholith

|
A very large pluton with steeply-dipping
walls, no known floor and a surface exposure in excess of 100 km2
|
Batholiths usually form from viscous
(silica-rich) granitic
melts and may in many cases represent megastocks beneath rhyolitic
calderas like those of the San Juan Mountains |
|
|
Sill

|
A sheet-like intrusion emplaced parallel to country rock bedding or
foliation planes
|
Sills require a low-viscosity magma and a
shallow level of emplacement. The unexposed sill beneath the Maroon Bells
elevated, hardened and grayed their Maroon
Formation rocks, which do not normally support such tall, angular
peaks. |
|
Dike

|
A sheetlike intrusion crossing
country rock bedding or foliation planes
|
Dikes surround many intrusions and often serve
as feeders for associated volcanoes. Radial dike patterns and dike swarms are
common. Even narrow dikes can conduct massive volumes of magma in
relatively short periods of time. Dike formation requires only that the
direction of least stress be horizontal. With an asthenosphere always
ready to melt and rise to the surface, it's a wonder there aren't more of
them. |
|
|
Vein
|
A small dike, often filled with pegmatite or aplite
|
Pegmatite veins are common ore bodies. |
|
Intrusive Map Units
On the Geologic Highway Map of Colorado, 1.7
Ga granites are marked "Xg" and mapped in light gray. The 1.4
Ga Berthoud and 1.1 Ga Grenville
granites are lumped under "Yg" in a darker gray. The Laramide
intrusions are tagged "TKi" and mapped in a dark purple, while the
mid-Tertiary intrusions are marked
"Tmi" in hot pink. The TKi units are pretty much confined to the Colorado
Mineral Belt; the Tmi units are also heavily concentrated there but have a
much wider distribution, including an occurrence at Spanish Peaks. Younger
intrusives under 20 Ma labeled "Tui" in stippled pink mainly occur
north of Steamboat Springs.
Top
Page Index
Sedimentary Rocks
Sedimentary rocks form from
-
the accumulation and consolidation of
loose surface materials, like sandstones and shales
-
the chemical precipitation of minerals from over-saturated waters, like evaporites
-
the remains or secretions of living organisms, like limestones
and cherts.
Because they're a lot more stable under surface
conditions than igneous or metamorphic rocks, sedimentary rocks now cover
two-thirds of the total area of the continents to an average depth of ~0.5 km
(1,800'). By volume, sedimentary rocks are about
two-thirds mudstones world-wide. In Colorado, the sedimentary cover ranges from absent over the
Precambrian cores of Laramide uplifts to 4.0 km (13,000')
thick at the west end of the Denver
Basin.
It will once again prove useful to categorize
sedimentary rocks, but bear in mind that there will always be transitional rock
types. Nature, in her devotion to
entropy, just loves to mix things up. This section will continue with the
dominant class of sedimentary rocks known as clastics. The following section will
describe the chemical sedimentary class—a very
different kettle of rocks indeed.
For more on sedimentary rocks, visit the extensive
and well-illustrated ^sedimentary
rock site by educator and geologist ^Lynn
Fichter. They're not as dull as you might think. For an introduction to
stratigraphy, the science of reading stacks of sedimentary rocks, see Formations
and Sequences below.
Clastic Sedimentary Rocks
Clastic sedimentary rocks form from clasts
(fragments) weathered or otherwise disaggregated from other rocks. A synonym for
clastic is detrital. Clastic
rocks can be
categorized in any number of ways, but most classification schemes key on both grain
size and composition, which together tell a
lot about the source rock, the depositional environment, and all the steps in
between. Clastic rocks are generally held together by the interlocking of grains
and by chemical cements,
which also add color. Sandstones,
shales and carbonates like limestone
account for over 95% of all sedimentary rocks because they're the most stable
rocks at
surface conditions. When the source rocks are volcanic, the sediments are
said to be volcaniclastic.
Clastic Sedimentary Classification Schemes
I can't improve on the sedimentary classification web sites developed by
geologist and educator ^Lynn
Fichter, so I'll just link his explanation of the ^QFL
(quartz, feldspar, lithic) naming system here. The QFL system takes into
account both texture and composition,
but its use is beyond the scope of this site.
Clastic Grain Sizes
Clastics can be subdivided and are often named
according to their average or dominant particle size according to the logarithmic Wentworth
Grain Size Scale summarized in the table below.
Wentworth Grain Size Scale
|
Clast
Type
|
Maximum
Size (mm)
|
ø (phi) scale
|
Gravel
If the stone contains angular clasts, it's a breccia;
if rounded, it's a conglomerate.
|
Boulder
|
n/a
|
-12
|
Cobble
|
256
|
-8
|
Pebble
|
64
|
-6
|
Granule
|
4
|
-2
|
Sand
|
Very coarse |
2 |
-1 |
| Coarse |
1 |
0 |
| Medium |
1/2 |
1 |
| Fine |
1/4 |
2 |
| Very fine |
1/8 |
3 |
| Mud
|
Silt |
1/16 |
4 |
| Clay |
1/256
|
8
|
Technical Note: The ø (phi) scale facilitates grain size statistics;
ø = -log2 mm. It's worth repeating that the Wentworth scale is about grain size, not composition.
Any particle in the 0.0625-4.0 mm range is sand, whether it's made of pure
quartz or feldspar or undisaggregated rock fragments. In the last case, we might
speak
of a lithic sandstone, but it's a sandstone nonetheless. The same applies to the clay
category, but here, size implies composition:
Few clastic sedimentary rocks composed of clay-sized grains will contain
anything other than clay.
Sedimentary
Rock Names Based on Grain Size
Common sedimentary rock names like sandstone, mudstone, siltstone,
and claystone come directly from their average grain sizes, as defined in
the Wentworth grain-size scale above. Thus, a rock with
grains averaging 0.375 mm across is a medium sandstone.
Shale, a
term synonymous with mudstone, includes any fine-grained clastic sedimentary
rock originally made of mud. Thus, siltstones and claystones are both shales. Pelite
is yet another name for mudstones. Wind-deposited silts bears the special name loess.
Of course, all size combinations are possible, including sandy siltstones and silty claystones.
The term wacke lumps together silty and shaley sandstones, which can be
hard to tell apart in the field. If a
gently bitten shale feels gritty between the teeth, it's one-third to two-thirds
quartz silt and may qualify as a wacke. If it feels smooth or creamy, it's at least two-thirds clay.
A
conglomerate is dominated by rounded gravel-sized clasts (> 2 mm),
while a breccia contains angular gravel. At right is the basal layer of the Fountain
Formation at Red Rocks Park, a moderately coarse conglomerate containing rounded
pebbles and granules up to ~50 mm across in a sandy matrix—a typical terrigenous
deposit. Since such deposits develop in alluvial fans, they're sometimes
called fanglomerates.
Grain Sorting and
Shapes
The standard deviation of grain sizes found in a clastic rock is a measure of its
degree of sorting, which in turn says something about the rock's porosity,
permeability and depositional history. In the field,
the standard deviation is half the size range that includes two-thirds of all
the grains. If two-thirds of all the grains in our medium sandstone above fall between
0.5 and 0.25 mm, the standard deviation is (0.50-0.25)/2 = 0.125 mm. Clastic
rocks deposited near their source tend to have poorly sorted grain sizes.
Well-sorted rocks are both more porous and more permeable than poorly sorted
rocks with the same average grain size. The degree of sorting is a measure of
the textural maturity of a clastic rock.
Grain shapes also tell a story. Grain shapes are usually described in terms
of their sphericity (the degree to which all dimensions are equal) and roundness
(the lack of sharp corners). Sphericity is often controlled by composition.
Grains can be rod-shaped, disc-shaped, or spherical. The degree of rounding
depends on grain size and hardness and on details of transport,
deposition and diagenesis.
Clastic Grain Compositions
Sedimentary rocks typically include grains of the following components:
Sedimentary Rock Compositions
|
| Component |
Constituents and Weathering Patterns |
| Quartz (SiO2) |
This stable end-product
weathers to nothing else. Unfortunately, quartz grains give away little of
the sediment's past. |
| Feldspar |
Since feldspars quickly weather to clay when exposed at the
surface, their presence in a sandstone like the Fountain Formation
indicates rapid burial and a nearby source. |
| Lithic
fragments |
Rock fragments weathering to quartz, clay or
carbonate |
| Mineral grains other than quartz, which
usually weather to clay |
| Matrix |
Silt- and clay-sized grains of any composition not
easily visible to the unaided eye. |
| Organic material |
Plant or animal remains often reduce to
elemental carbon during diagenesis. |
Most
sedimentary classification schemes key on the relative proportions of quartz,
feldspar, lithic fragments and matrix. The ^QFL
(quartz, feldspar, lithic) naming system is such a scheme.
Ever since plants became abundant in the
Silurian around 420 Ma, it's been hard to find a clastic rock devoid of
organic content, but organics usually don't figure explicitly into the
classification of clastic rocks, however important they may be to petroleum
geologists. At
right, organic carbon darkens some of the layers in the otherwise light-colored South Platte sandstone member
of the mid-Cretaceous Dakota Group.
Note that grain size and composition aren't entirely independent. Quartz grains
battered down to silt size go into suspension in flowing water, where they are
largely immune from further diminution and rounding by impact and abrasion. At
maturity, quartz grains tend to stabilize at silt size with some residual angularity.
Clastic Sedimentary Structures
Sedimentary structures reflect depositional heterogeneities at scales larger than texture. They
provide valuable and often quite specific information about the depositional environment.
Bedding
 |
I-70 road cut
|
Bedding
is the most obvious example of structure. Bedding planes reflect some change in
the depositional environment, whether it be
-
the texture or composition of material
deposited—e.g., sand, mud or lime
-
the method of transport and deposition—e.g., wind
or water, fast or slow currents, river or beach bar
-
groundwater chemistry—e.g., rich or poor in
dissolved cement minerals or organic acids
The famous I-70 road cut west of Denver (above) exposes obvious bedding in Cretaceous and Jurassic
sediments of the Dakota Group, Lytle
Formation and Morrison Formation,
which go from left to right in this picture.
Graded
Bedding
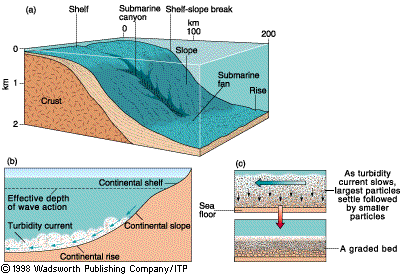 |
Turbidity currents and turbidites
|
Graded
bedding with grain size coarsening toward the base is usually seen in turbidites—marine
sediments deposited by turbidity currents in fans on or near the continental
rise. As the current slows, the larger, heavier grains drop out first. The cycle
repeats with each new turbidity current. The diagrams at right show the basic
features of this process, which operates along passive
continental margins and magmatic arcs as
well.
Cross-bedding
 |
Cross-bedding
|
Cross-bedding
is a common structure in sandstones and siltstones, whether water- or wind-laid.
Sand dunes, beaches, tidal flats and river bars all produce cross-bedding of
varying steepness and scale, but individual beds usually dip down the current
that deposited them. The 1.7 Ga Coal Creek
quartzite still shows delicate cross-bedding after all these years.
Ripple Marks
 |
Ripple marks
|
Ripple
marks are the first bedforms to appear when fluid (air or water) flow
becomes fast enough to transport sand. Symmetrical ripple marks like those
exposed in the Dakota sandstone on
Dinosaur Ridge (right) tend to form in lakes and on beaches, where water
currents are bi-directional. Asymmetric ripples formed by streams or winds are
steeper downstream or on the lee side.
Other
structures
 |
Worm burrows
|
Other
structures include concretions, mud cracks, bioturbation (destruction of
bedding planes by burrowing animals), imbrication (stacking of flat grains at an
angle to the bedding plane, with dip upstream) and flute casts (bed gouges dug
up by passing turbidity currents), and trace fossils like the worm
holes at Dinosaur Ridge (right).
Porosity and Permeability
The voids between individual grains in a clastic sedimentary deposit may be reduced
during compaction, but they're rarely eliminated. They provide opportunities for
cementation of the sediment grains and for fluid transport and storage—issues
near and dear to the hearts of petroleum geologists and hydrogeologists.
Porosity
Porosity is the ratio of
residual void volume to total rock volume. In a sandstone, greater
angularity and better sorting of the sand grains both increase porosity, while
tiny clay grains reduce porosity by collecting in and plugging up the voids.
Cements also reduce porosity. Sedimentary rocks with high porosities can host a
variety of fluids, from ground water to magmatic fluids to petroleum to natural
gas.
Permability
Permeability, the ease with which a fluid can move through the pore
spaces of a rock, is a different matter, and clay content once again enters the
equation. Clay grains have charged surfaces and high surface-to-volume ratios.
These properties retard flow by increasing the attractive forces between pore
walls and contained fluids.
The
stiff, angular quartz grains and the low clay content in a sandstone like the
South Platte member of the Dakota Group
of eastern Colorado (right) give it high porosity and permeability.
Petroleum and natural gas generated in the underlying highly organic Benton
shale (the source rock) migrate upward into the South Platte (the reservoir rock), where they are free to flow and collect in folds that serve as
traps.
Clastic Cements
How well consolidated a sedimentary rock becomes
depends on
-
The strength of its constituent materials
-
How well individual particles or crystals
interlock under compaction with burial
-
How well individual particles become cemented together by
precipitates from the fluids permeating their pore spaces.
Since cements play a critical role in the color and
durability of sedimentary rocks, they deserve a little detail.
Water-Borne Cements
Water is by far the most common pore-space fluid found in sedimentary
deposits. Water rich in dissolved iron oxides, calcite (calcium
carbonate) or silica (silicon dioxide) can deposit these minerals as cement
between sediment grains. Well-cemented sedimentary rocks like the Kayenta
Formation (right) tend to be resistant to erosion, but if they ever encounter
pore fluids capable of dissolving their cements, all bets are off.
Silica (SiO2) is
the least soluble and therefore the most durable cement, but it's rare
outside sandstones with 90% or higher quartz contents. Hematite (Fe2O3)
is another durable cement notable for the wide range of reddish hues it imparts.
Calcite (CaCO3) is the most
soluble and the least durable of the common cements, particularly in the presence of acidic ground
water. When the cement dissolves, the sedimentary rock becomes friable
and eventually disaggregates on exposure.
Magmatic Cements
| Mineral-rich magmatic fluids containing
water, silica and other minerals can also cement
sedimentary rocks, but the heating and chemical alterations they bring
usually push sedimentary country rock into the metamorphic
realm. Nearby igneous intrusions left the Late
Paleozoic sedimentary rocks of the Maroon
Formation in this transitional state at the Maroon
Bells (upper right) in the Elk Range.
Magmatic fluids emanating from another Elk Range intrusion altered the
Maroon Formation sediments now cropping out in the rugged peaks around Cathedral
Lake (lower right) into a tough, lustrous frankly metamorphic
gray-green rock called hornfels. |
 |
|
|
 |
|
|
Sedimentary or Metamorphic?
At what point altered sedimentary rocks like those at the Maroon
Bells become metamorphic has always been controversial, but there are some widely
accepted conventions.
Clastic Colors
By
themselves, end-stage sediments aren't very colorful. Quartz grains are usually
clear or gray, most clay minerals are faintly green at best, and carbonates tend to be
white, gray or even faintly blue.
Yet clastic rocks show an extensive palette of colors, sometimes subtle,
sometimes vibrant, including white, gray, black, buff, brown, green, yellow, orange, red and even
purple and blue. Where do the colors come from?
The short answer is cement, and it takes only a few
percent of tinted cement to make a colorful rock, as in the red basal
conglomerate of the Fountain
Formation west of Denver (above).
Brown, red, orange, yellow and purple hues come from mixtures of iron
compounds in pore spaces. Hematite (Fe2O3) is red; iron
hydroxide is brown; and iron silicates are green, as are most clay
minerals. The iron ultimately comes from weathered ferromagnesian minerals
common in igneous and some metamorphic
rocks. Hematite cements are common in terrigenous
sediments, especially in arid climates, as seen in the Triassic
redbeds exposed at right in the north wall of Maroon
Creek Canyon in the Elk Range.
The
grays through blacks come from free carbon derived from organic materials
incorporated into the sediment, as in the coaly layers within the Dakota
sandstone at right.
Blue comes from cements containing lawsonite or glaucophane—both
high-pressure, low-temperature blueschist minerals found in rocks
previously taken to depths of 20 km or more and brought back up again in a subduction
zone setting. Some limestones can also take on a bluish cast, but I have no idea
why.
Continental Shelf Deposits—Sedimentation 101
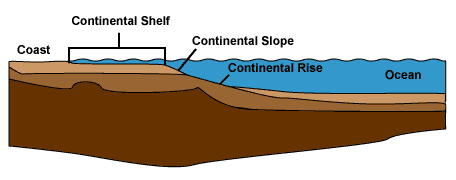 |
Passive continental margin in cross-section
|
Passive continental margins like the
Atlantic coast of North America tend to acquire submerged shelves extending tens to hundreds of
kilometers offshore to end at the continental slope. The shelves typically
consist of
flat-lying fine-grained end-stage sediments
accumulating to great thicknesses over faulted
continental crust. Some shelves, like those bordering the Red Sea, advance out
onto oceanic crust as well.
In many ways, continental shelves are the
simplest of depositional environments, so we'll start by tracing their
constituents through all five phases of sedimentation. These phases occur
in all types of sedimentation, but they are most developed in the shelf setting.
Phases of Sedimentation
|
| Phase |
Definition |
|
Weathering |
Breakdown of solid rock, chemically or
mechanically |
|
Erosion |
Mobilization of weathered rock by water, ice
or wind |
|
Transport and Sorting |
Hauling of sediment from source to deposition
site, sorting along the way according to differences in grain
transportability (size, shape, density, etc.) |
|
Deposition |
Accumulation of sediment released from the
transport system |
|
Diagenesis |
Compaction, heating and cementation of grains
ending in lithification (rock formation) |
Climate strongly controls all five phases, but bear in mind that climate can
change drastically between source regions and and depositional environments—as
it does, say, between the glaciated alpine peaks and subtropical shores of New
Zealand.
Weathering
Once exposed at the surface, rocks begin to
weather—i.e., to break down into disaggregated sediments due
to inherent chemical and mechanical instabilities. Examples of chemical weathering include
-
Breakdown of the ferromagnesian minerals in
crystalline rocks into clay minerals and red
hematite (Fe2O3)
-
Breakdown of all other common non-quartz igneous
minerals into clay minerals and dissolved silica
-
Dissolution of limestones and sedimentary cements by ground water laced with organic acids
-
Oxidation of iron-bearing clay minerals to form
aluminum-rich laterite (red clay) soils in hot, wet climates
 |
Granite domes
|
Frost-wedging, the most important form of mechanical weathering,
depends on the fact that water expands by ~9% on freezing and operates anywhere
freeze-thaw cycles repeat through the winter—particularly on rock faces in
high mountain regions with wet, temperate climates. When water filling a crack
freezes, the expansile force generated is often sufficient to open the crack a
bit. If the crack parallels the surface, repeated wedging will eventually split
the overlying slab off the rock face to fall into a talus slopes below. Abrasion of bedrock by bedload sediments in
a swift mountain stream, or by other rocks embedded in the foot of a glacier, is another
common form of mechanical weathering. A final form is pressure release,
wherein rocks formed at high pressure split apart spontaneously on dispatch to
the surface due to internal stresses. The granite domes of ^Rocky Mountain National
Park (right) owe their shapes to the process of exfoliation,
which combines pressure release and frost-wedging on a large scale.
Chemical weathering is more effective at dismantling rocks than mechanical weathering by orders of
magnitude, but the two are often synergistic. As mechanical weathering reduces
clast size, it increases the total surface area susceptible to chemical attack. Conversely, as chemical weathering dissolves
its cement, sedimentary rock
becomes more easily disaggregated by mechanical weathering processes.
Erosion
 |
Royal Gorge
|
Erosion is the mobilization of weathered
rock, usually by a moving fluid, be it creeping glacial ice, flowing water or
wind. Erosion feeds weathered rock into the transport system(s) made available
by topography and climate. Glacial ice is by far the most effective erosive agent, and wind the
least. Short of glaciers, most of the work
of erosion is done by bank-full streams and 100-year gully-washers, not by
average weather. These floods allow streams to carve imposing canyons without
glacial help. A steep gradient and an ample supply of water and bedload helped
the Arkansas River cut Royal Gorge (right).
Transport and Sorting
Shelf sediments typically travel great distances
to their resting places, weathering all the way. By deposition time, they've
usually been reduced to stable end-products—quartz sand, clay mud and dissolved
carbonate. But flowing water transports sand, clay and lime quite
differently, and the differences lead to sorting.
Since the larger sand particles tend to bump and roll along
the bottom until they go back into suspension at silt-size, they tend
to settle out first as water velocities diminish in shallow, near-shore
settings. The much smaller,
lighter, flatter clay particles are carried in suspension and drop out only in
much deeper, quieter water.
The calcium carbonate is carried in solution and comes out only when
actively pulled out by shell-building fresh water or marine organisms like
corals and oysters. These light-dependent organisms are usually most abundant in
the clear water just beyond the clay deposition zone but under 50 m in
depth.
Air masses
can spread fine clay dust across oceans, but wind can carry sand only so far up
the dune face or down the back side.
Dissolved carbonate aside, flowing ice can carry anything it pleases, from mud
to house-size boulders. That's why glacial deposits are so poorly sorted.
Note that stream transport can't round quartz
grains completely, but surf and wind can come close.
Deposition
[diagram coming]
|
Marine sediments vs. depth
|
When streams lose their momentum at lower elevations, they drop their loads
in roughly this order: Large clasts, small clasts, sand and mud (silt and clay). The
lime stays in solution until the water reaches the right ecosystem. This hierarchy
effectively sorts sediments, first by size and density, and then, at end-stage,
by composition. Thus sandstones are usually near-shore and stream bed deposits,
clays are usually flood plain or deeper water deposits, and limestones are
deeper still. Mixed sediments—like sandy or limey mudstones (the latter marls),
or muddy or limey sandstones, or sandy or muddy limestones—are always possible
under the right conditions.
Diagenesis
The alterations a sediment undergoes after deposition but short of
metamorphosis are lumped under the term diagenesis. Diagenesis is
generally taken to operate below 150-200°C, which may come at depths of 3-10
km, depending on the local geothermal gradient. Above those
temperatures, metamorphic signatures like foliation and the recrystallization of
quartz appear.
Lithification, the formation
of rock from sediment, is perhaps the most important outcome of diagenesis. The
table below lists the most common
diagenetic processes.
Diagenetic Processes Leading to the Lithification of Sediments
|
|
Compaction
|
Expresses fluids, increases grain-to-grain contact
|
|
Heating
|
Increases ductility of all grains, leading to better interlocking; helps ground
water pick up more cement minerals
|
|
Cementation
|
Fills pore spaces, partially or completely, increasing effective
grain-to-grain contact
|
Let's follow a shelf sand deposit through diagenesis to lithification.
Similar things will happen to other sediments. The
sand will compact under their weight of overlying deposits. Compaction increases the surface area of
intimate grain-to-grain contact, but not so much with quartz grains, which are
rigid and angular and tend to meet only at points. More ductile lithic
grains (e.g., of
schist or shale) will squeeze
into pore spaces to some extent while the stiffer
quartz grains hold their ground. The more angular the stiffer particles are,
the less the ductile grains can flow. Fluids, most notably ocean water, ground
water or petroleum, will circulate through the residual pore spaces, depositing
any over-saturated minerals they might contain as cement.
The cement increases the strength of the forming rock by further increasing the
total surface area of effective grain-to-grain contact. At any point in the
process, the residual pore spaces can hold fluids which may add or dissolve
cement.
Heating (up to 150°C) is usually the most important agent in diagenesis. Increased
pressure due to burial usually acts only through compaction. Pressure-related
changes in mineralogy occur only in metamorphic
settings.
Pelagic Deposits
[photo coming]
|
Pelagic chert
|
Shelf sediments seldom reach the deep ocean floor of the abyssal plains.
Transport mechanisms are generally lacking, and sinking carbonate skeletons
dissolve before reaching the bottom.
In pelagic (deep open-ocean) settings below the carbonate
compensation depth, radiolarian cherts dominate the
sedimentary veneer covering the igneous rocks of the oceanic crust. Insoluble
silica-based radiolarian skeletons rain down from the sunlit surface layer onto
the
ocean floor to make ~1 mm of chert per 10 Ka at any depth. With burial and
heating, the skeletons recrystallize to form thin (10-50 mm) beds of nearly pure microcrystalline
quartz
separated by even thinner beds of shale chemically segregated from the silica
during recrystallization. The shaley partings derive from microscopic clay dust particles
dropped over the oceans by global winds passing over the continents. Such sequences are also known as pelagic,
bedded, or ribbon
cherts. No other
sediment sources are available to the oceanic
crust of the abyssal plains.
In the field, pelagic cherts appear as bedded or ribbon
chert, sometimes in layers well over 50 m thick. The sporadic horizons of small
chert nodules found in limestones like the Paleozoic limestones of western Utah
around Canyonlands National Park represent chemical segregations of minor inputs
of radiolarian silica in a shallow biogenic depositional environment dominated by
carbonate inputs. Any chert found in limestone must have been deposited above
the carbonate compensation depth.
Deposits Left by Transgressing and Regressing Seas
As a transgressing sea like the Cretaceous Interior
Seaway invades a landscape, it first lays down beach and barrier bar
deposits. As it deepens, it deposits clay over the sands. As it deepens further,
carbonates accumulate on the bottom as corals and shellfish set up shop in the clear
water. If the sea
regresses, muds bury the limes, then sands bury the muds as the water shallows.
Terrigenous (dry land) strata then bury the entire marine sequence once the
water is gone.
On
burial and lithification, such depth-sorted sediments form
a sandstone-shale-limestone-shale-sandstone sequence marking the coming and
going of the sea. A Cretaceous to
Paleocene transgressive-regressive sedimentary sequence crops out throughout
Colorado, notably at the Dakota Hogback
(above right) west of Denver. There, the marine strata are, from oldest to youngest,
are the transgressive Dakota sandstone, the Benton
shale, the Niobara limestone, the Pierre
shale, and the regressive Fox Hills and Laramie
sandstones. The terrigenous Arapahoe and Denver
Formations still cover the younger portions of the sequence. The Niobara hogback
is long lost to quarrying.
Terrigenous Deposits
 The marine deposits we've examined so far make up a substantial
fraction of
the sedimentary rocks found in Colorado, but terrigenous sediments
deposited on dry land are also strongly represented. With few exceptions, the
voluminous Late Paleozoic Fountain, Maroon
and Hermosa sediments shed from the Ancestral
Rocky Mountains, most of the pre-Dakota
Mesozoic strata, and the
sediments shed from the current Rockies during and after the Laramide
orogeny were transported and deposited
on land by streams. Lacustrine (lake bed) deposits like the Eocene Green
River Formation are transitional in this regard. The marine deposits we've examined so far make up a substantial
fraction of
the sedimentary rocks found in Colorado, but terrigenous sediments
deposited on dry land are also strongly represented. With few exceptions, the
voluminous Late Paleozoic Fountain, Maroon
and Hermosa sediments shed from the Ancestral
Rocky Mountains, most of the pre-Dakota
Mesozoic strata, and the
sediments shed from the current Rockies during and after the Laramide
orogeny were transported and deposited
on land by streams. Lacustrine (lake bed) deposits like the Eocene Green
River Formation are transitional in this regard.
The table below lists some important terrigenous depositional environments,
derivative Colorado rocks and modern examples. Formations listed together
accumulated in laterally equivalent or persisting environments; those listed
under separate bullets represent recurrences of the same depositional
environment.
Terrigenous Depositional Environments
in Colorado's Past
|
| Depositional Environment |
Derivative Sedimentary Rocks in Colorado |
Modern Example |
|
Alluvial fan |
|
Basin and Range |
|
Floodplain |
|
Lowlands surrounding the Gulf of Mexico |
|
Coastal mudflat |
|
Southern shores of the Persian Gulf |
|
Desert dune field |
|
Sahara Desert |
Redbeds
Hematite
cements released by the weathering of
ferromagnesian source rocks grace many terrigenous deposits with a range of
reddish hues—hence the term redbeds. An arid depositional climate seems
to help bring out the red, and such environments were the rule rather than the
exception in Late Pennsylvanian through mid-Jurassic Colorado. At right, Late Paleozoic redbeds of the Fountain
Formation crop out in Red Rocks
Park near Denver. The photo at the top of this
subsection shows Triassic
redbeds exposed in Maroon Creek Canyon in the Elk
Range. Marine sediments are less likely to show such colors.
Clastic Sedimentary Map Units
In keeping with the worldwide dominance of clastic rocks at the surface, most
of the bedrock units shown on the Geologic Highway Map of Colorado
are largely or entirely composed of clastic sedimentary rocks, particularly the
Cenozoic strata. Colorado is under quota in this regard, however, because
erosion has already stripped the sedimentary cover from the summits of many of
its Laramide uplifts.
Chemical Sedimentary Rocks
[photo coming]
|
Pelagic chert
|
Sedimentary rocks precipitating from an
over-concentrated solution (say, from the dregs of an evaporating sea) or
manufactured by organisms are said to be chemical, and the latter, biochemical.
They're generally quite different than their clastic
brethren and constitute less than 5% of all sedimentary rocks. Almost always
water-laid, chemical rocks supply
their own cement to any water able to circulate through their
pore spacess,
but they're generally low in both porosity and permeability.
Petroleum geologists don't go looking for oil in chemical rock formations, but
chemical rocks can seal off petroleum traps in underlying strata.
Chemical rocks typically weather by dissolving into
surface or ground waters with or without the aid of mechanical weathering. Ground
water is more often than not acidified by organic materials (less often by acid rain),
and that makes it a highly effective
solvent for chemical rocks, especially the carbonates.
Given their solubility, chemical sedimentary rocks are understandably much less
common in outcrop than their clastic counterparts, but chert, a
tough chemical rock made of very stable
microcrystalline quartz, is one of the most resistant of all surface rocks, and limestones
can form spectacular landscapes in the right setting—e.g., the famous ^karst
towers of Guilin, Guanxi Province, south China.
Carbonate Rocks
Carbonate rocks are by far the most important
of the chemical rocks—both by volume and by virtue of their critical role in
the planet's carbonate cycle, which among other things, controls the carbon
dioxide content and therefore the green-house properties of the atmosphere. Most
carbonate rocks are biochemical.
Limestone
Limestone
forms directly from the compacted calcium carbonate skeletons and shells of
corals and other calcareous organisms. It takes many, many different forms,
depending primarily on the depositional environment. Physicochemical processes
related primarily to decreasing temperature and increasing dissolved CO2
concentration with depth in ocean water force precipitated carbonate minerals
back into solution below the carbonate compensation depth (CCD), which
today is ~4 km down. Since marine limestones can only form above the CCD,
geologists find them a useful if rather coarse paleodepth indicator.
Dolomite
[photo coming]
|
Leadville limestone and dolomite
|
Dolomite, another
important biochemical carbonate rock, is a chemical variant of limestone in
which some of the calcium atoms are replace with magnesium. Interestingly, in
Paleozoic and earlier times, dolomites outmassed limestones by about four to
one, but dolomite comprises only a tiny fraction of the carbonate rocks forming
today. No one knows why. Despite the name, the Mississippian
Leadville Limestone of Colorado mining fame is mostly dolomite.
Marl
Marls are mixtures of clays and carbonates with at
most minor amounts of quartz. Weathered marls make challenging soils for growers
and civil engineers alike.
Cherts
[photo coming]
|
Pelagic chert
|
Cherts are very hard, highly resistant and often colorful
chemical rocks of microcrystalline silica that tend to form topographic highs
wherever they crop out in bedded form. Weathered bedded chert can look like a
fine-grained sandstone from a distance, but fresh surfaces are lustrous and
slightly translucent. Chert colors range from pure white, gray and black to yellow, green, rust
and red, depending on the impurities involved. Chert nodules ranging up to
meters across and weathered or pried from Paleozoic limestones have been an
important raw material for the native stone toolmakers of the US Southwest, who
valued chert's exceptional hardness and conchoidal fracture habit. Pelagic
cherts provide geologists with reliable markers of deep ocean floor
environments.
Organic or Biogenic
Cherts
Most chert forms indirectly from planktonic
debris—specifically, from the accumulated
siliceous (silica-based) skeletons of radiolaria and diatoms after recrystallization of their
silica on burial heating to temperatures of 80-100°C. [??] Logically, chert
formed in this manner might be classified as biochemical, but
because of the required recrystallization step, this transitional
rock type is classed as chemical by
convention. Pelagic radiolarian cherts form
on the ocean flow below the carbonate compensation depth.
Diatomaceous ribbon cherts form in carbonate-starved lake settings under certain
conditions.
In shelf environments, cherts usually appear as small nodules
chemically segregated and recrystallized from the minor biogenic silica trapped
in massive limestones. For reasons that remain unclear, the nodules tend to line
up along limestone bedding planes.
Inorganic Cherts
Truly chemical or inorganic cherts do form
occasionally in lakes that become severely alkalinized on
a seasonal basis—e.g., by blooms of
photosynthetic algae or by winter drainage from nearby sodium carbonate lavas (carbonatite).
Normally undersaturated dissolved silica becomes oversaturated at high pH (>
10) and precipitates out as a silica gel that crystallizes directly to chert
with burial. Colorado's Morrison
and Green River Formations contain
inorganic cherts.
Evaporites
Evaporites are perhaps the purest form of
chemical rock. They typically appear in the rock column as massive gypsum (CaSO4)
and rock salt (halite, NaCl) deposits left behind by an over-saturated body of
natural water trapped in a restricted basin with a limited
refresh rate. Evaporites usually form in arid climates with a high evaporation
rate and little rainfall. ( Think Bonneville Salt Flats.) In the geologic
record, they've most often formed in the planet's desert belts at 30-40°
paleolatitudes.
Since evaporites are highly soluble and easily
eroded, they're not often found at the surface, but the razor-sharp halite
foothills of the west slope of the Zagros Mountains of Iran are a notable
exception. Gypsum is the least soluble of the common evaporite minerals and
therefore the most likely to be found in outcrop.
Late Paleozoic evaporites of Ancestral
Rocky Mountain provenance are common in the Maroon and
Paradox Basins of central and southwestern Colorado, respectively. Due to their unusual chemical and mechanical
properties, evaporites
make for odd topography, like the chaotic Pennsylvanian black shales disrupted
by rising gypsum bodies along the Colorado River between Dowd and Wolcott on
I-70.
Chemical Sedimentary Map Units
From both an outcrop and economic standpoint, one of the most important
chemical sedimentary units in Colorado is the Mississippian Leadville
Limestone lumped into the dark purple Early Paleozoic "MDOC" unit
on the Geologic Highway Map of Colorado. The
thick evaporites of the Maroon
and Paradox Basins are mapped under the light blue "PP" unit. The
inorganic cherts of the Green River and Wasatch Formations will be found within
the pale yellow "Tl" units of western Colorado.
Top
Page Index
Metamorphic Rocks
Igneous
and sedimentary rocks are stable only at the conditions under which they
became rocks. Since most rocks form at some depth below the surface, their
equilibrium temperatures (T) and pressures (P) are usually higher than those
found at the surface—sometimes much higher. If uplift and erosion return a
high-T, high-P granite to the low-T, low-P surface, it will adjust its new
environment via chemical and physical weathering, as
we saw in the discussion of sedimentary rocks above.
But what about the opposite scenario? If a continent-continent
collision deeply buries a shale stable under low-T,
low-P near-surface conditions, its
elements will reshuffle into a new mineral
assemblage better suited to the high-T, high-P conditions found at
depth. Along with the chemical adjustments come textural
changes stemming from recrystallization along preferred
orientations, microfracturing, and even plastic flow. These alterations go beyond
diagenesis
to enter the realm of metamorphism. The shale, the parent rock,
can give rise to many different metamorphic products, depending on the conditions
involved. If those products return to the surface, they can yield important
clues regarding where the rock's been and what kind of rock it might have been
when it started its journey.
This section will explore the many faces of metamorphism. For more on
metamorphic rocks, visit the extensive and
well-illustrated ^metamorphic
rock site by educator and geologist ^Lynn
Ficther.
Eclogite, Subduction's Whip
Lest you think that metamorphism is just something that happens to rocks that
stray too deep, consider the story of a certain high-pressure garnet-bearing
metamorphic rock called eclogite. The Ligurian Alps and
Ring Mountain in northern California are among a handful of places in the world
offering substantial in situ exposures. Eclogite isn't rare—by now, the upper
mantle is riddled with the stuff. It just takes an extraordinary upheaval to
bring it to the surface.
But all mantle solids are rare at the surface. What's so special about
eclogite? Simply this: Subduction drives all of plate tectonics, and eclogite is
its whip. In fact, a critical repositioning of the depth of eclogite formation
by the cooling of the earth was an important precursor to the onset of plate
tectonics at ~2.0 Ga.
Continuing top-down cooling of the
upper mantle produces gravitationally unstable
oceanic lithosphere that progressively cools, thickens, stiffens, imbibes
water and becomes more dense through cooling and phase changes as it rafts away from its
spreading center
of origin. Not far from its spreading center, the oceanic lithosphere (slab
for short) is
already denser and stiffer than the hot asthenosphere on which it
rests. Flexural support and lateral
compression by surrounding lithosphere allow it remain topside for a while
despite the density inversion, but eventually, all overdense slabs buckle
downward and sink subvertically into the upper mantle along
convergent plate boundaries known as subduction
zones. The rate at which gravity pulls down the slabs sets the pace for the
rest of the system—continental drift, arc magmatism, the works.
That's where eclogite comes in. Once the falling slab reaches a depth of ~60
km, the basalt and gabbro making up its crust rapidly convert to much denser eclogite—denser
even than the upper mantle rock below the asthenosphere. Like a lead sinker on a
fishing line, the eclogite hastens the slab's fall. Moreover, water released
into mantle wedge overlying the slab as
a chemical byproduct of the eclogite transition eventually triggers the partial
melting that fuels arc magmatism, an
important pathway for the cooling of the earth and the recycling of upper mantle
into continental lithosphere.
So eclogite helps the world churn and burn. It'd be a much duller place
without the metamorphism that produces eclogite.
Schist Happens—The
Shale Pathway
To make the metamorphic process less abstract, and less exotic, let's follow a
common shale through
its metamorphosis as both temperature and pressure progressively increase. In this
example, shale is the unaltered starting point or parent rock for the
metamorphic process.
Shale Under Increasing Temperature and
Pressure
|
| Mudstone Derivative |
Required Alteration |
Field Indicators |
^Shale
|
|
|
^Slate
|
- Recrystallization of clay minerals to platy chlorites
- Reorientation chlorite crystals perpendicular to the local
pressure (stress) gradient
|
- Microscopic grains
- Distinct, smooth slaty cleavage, often at an angle to the
original bedding plane
|
^Phyllite
|
- Increased grain size, particularly in mica and chlorite minerals
|
- Microscopic grains
- Slaty cleavage with a distinct sheen
- Occasional tiny porphyroblasts
|
^Schist
|
- Further increase in grain size
- Chlorites alter to intermixed micas, quartz, feldspars and
amphiboles
- Formation of porphyroblasts, often of metamorphic minerals like
garnet, staurolite or cordierite
|
- Macroscopic grains
- Larger porphyroblasts
- Schistosity (irregular cleavage due to parallelism of platy
and elongate macroscopic crystals)
|
^Gneiss
|
- Segregation of light felsic (quartz and feldspars) and dark
mafic
minerals (micas, pyroxenes, amphiboles) into distinct layers
- Loss of schistosity
|
|
^Migmatite
|
- Partial melting, usually starting in the less refractory light-colored
felsic layers
|
- Wavy banding and other flow structures with igneous features in the light-colored layers
|
Table Note: The external links lead to illustrated descriptions of
metamorphic rocks at Lynn Fichter's Metamorphic
Rocks site.
Quartzite is Forever
 |
Highly recrystallized quartzite,
Quartzite Ridge, Eldorado Canyon State Park
|
 |
Less recrystallized Coal Creek quartzite, Golden Gate Canyon State
Park
|
Under increasing temperature and pressure, a mature quartz sandstone would have evolved quite differently than the shale
followed through the regional metamorphic process in the table above. After recrystallizing into a ^quartzite at
relatively low temperature and pressure, it would have resisted all further changes short of melting.
In the field,
quartzites and well-cemented pure quartz sands can be hard to tell apart, but a
hammer and a hand lens can help: Fractured
quartzites generally break through grains, sandstones between grains.
The Range of Metamorphism
When alterations due to increased pressure-temperature (PT) conditions or
permeating fluids exceed those associated with diagenesis,
the rock becomes metamorphic. The higher the temperature or pressure, the
higher the grade of metamorphism.
Lower Limit at 150°C
Practically speaking, the boundary between diagenesis and metamorphism is hard to define. Since
elevated temperatures are much more effective than elevated
pressures in altering rocks early on, it's common to define the lower limit of metamorphism in terms
of a temperature threshold, usually 150°C. Under typical geothermal gradients,
the temperature reaches 150°C at a depth of 3-4 km and a pressure of 0.5-1.0 k bar.
Upper Limit At the Onset of Melting
The upper limit of metamorphism is the onset of significant melting,
which occurs at 650-800°C for most crustal rocks. Such temperatures are common
in the lower crust near the Moho (the
crust-mantle boundary) but can also occur in the upper
crust in conjunction with magmatism. The presence of water, whether free in
pore spaces or bound in hydrated minerals, significantly lowers the PT
conditions needed to melt nearly any rock. Water is scarce at lower crustal
levels, where pore space is scant and anhydrous minerals are favored, but it
becomes progressively more abundant at middle and upper crustal levels.
Partial melting under extreme PT conditions is called anatexis.
It's more often the result of tectonism (regional metamorphism) than magmatism
(contact metamorphism) because magmas don't generally carry enough excess heat
to melt significant quantities of surrounding rock and remain molten at the same
time.
Migmatites, rocks with both igneous and metamorphic
features, are fairly good upper limit markers in the field. They always reflect
extreme PT conditions, but not all migmatites are anatectic.
Special PT Signatures
Temperature and pressure usually rise together during regional mountain-building,
the most common cause of metamorphism. The same is true of deep burial in a
subsiding sedimentary basin like the Denver basin on
the east flank of the Rockies.
But other telling PT combinations are observed. Normal subduction
zones, for example, offer unique high-P, low-T environments where otherwise
rare blueschists
containing minerals like lawsonite and glaucophane can form. In the field,
blueschists serve
as reliable signatures for subduction tectonics.
Igneous
intrusions, on the other hand, tend to create high-T, low-P metamorphic conditions in surrounding country rocks,
which can also be altered by permeating magmatic fluids. In the field, the
characteristic alterations can point to an otherwise unsuspected intrusion
nearby. Closing backarc basins at collisional
subduction zones represent a special case here. These very hot orogens
contain high-T, low-P environments capable of generating granulites.
Under the Hood
Rock go metamorphic for many reasons. Mountain-building is the most common
cause, but igneous intrusions, faulting, deep burial in sedimentary basins and a
variety of ocean floor processes
are also effective in altering rocks beyond the limits of diagenesis.
At rock level, pressure, heat, and fluids do the actual work of metamorphism.
Pressure
Pressure increases come with sedimentary and tectonic burial and with other
tectonic stresses. Elevated
pressures foster chemical change by forcing pre-existing grains into more
intimate contact and by favoring the formation of denser reaction products.
Pressure gradients prod crystals with elongate (rod- or disc-like) forms to
regrow
preferentially in directions perpendicular to the gradient. High partial
pressures of volatiles like water and carbon dioxide
can influence the course of metamorphic reactions and, in the case of water, can
reduce the melting points of most rocks. Elevated lithostatic pressures also
reduce melting points, with or without water.
Temperature
Temperature increases with burial, sedimentary or tectonic, and also with nearby magmatism.
Rising temperatures encourage chemical adjustments by destabilizing existing
minerals and by accelerating the diffusion of reactants. Elevated temperatures also favor the formation of
refractory minerals. At high temperatures, grain boundaries dissolve, cracks "heal",
previously segregated minerals come together and react, both in solid phase and
through fluid phase intermediaries,
and new products crystallize out in residual pore spaces.
The overall effect metamorphic heating is
to form new grains of larger and roughly equal (equigranular) size with
or without chemical change. In the presence of a pressure gradient, for example,
high temperatures mobilize mineral molecules at high-stress contact points and
allow them to recrystallize in low-stress pore spaces, effectively reorienting
the crystals as the gradient dictates without altering their chemistry.
Temperature-driven isochemical
recrystallizations tend to dominate metamorphism at low grades, as in the transformation
of shale to slate.
Fluids
Fluids enter the fray via groundwater, seawater, magmatic emanations and the release of volatiles.
Depending on the PT field involved, pore-space fluids can exist in liquid or
vapor phases and
can influence chemical and physical reactions in a number of ways. Water
transforms anhydrous into hydrous mineral forms and lowers the melting point of
the former even if hydration reactions don't occur. High pressures make
volatiles more reactive by increasing their partial pressures. High temperatures
make everything more reactive.
Types of Metamorphism
Metamorphic products vary with the PT field applied and with the presence of
fluids. Below are the most common types.
Regional (high-T, high-P) Metamorphism
Earth processes leading to metamorphism tend to involve large areas.
Compressive mountain-building is the prime example, but subduction
zones and regional magmatism bring metamorphic change to large regions as
well. Regional metamorphism typically brings elevations in both temperature and
pressure, sometimes to extremes. Aside from the loss of volatiles, regional
metamorphism is usually isochemical, meaning that the metamorphic rocks and
their parents retain very similar elemental
compositions. However, the products of regional metamorphism tend to become more
and more granite-like at high grades. This type of metamorphism is also known as
Barrovian after the pioneering British metamorphic geologist George
Barrow.
Contact (high-T, low-P) Metamorphism
 |
Contact metamorphism
|
Contact metamorphism is a high-T, low-P alteration of the country
rocks surrounding an igneous intrusion. A depth of 6 km
and a pressure of < 1.5 kbar would be a common scenario. Hardened,
reddened (oxidized) country rocks mark the thermal aureole or contact
zone ("bake zone"), which can extend for meters to kilometers
around the intrusion.
In silicate country rocks, the resulting metamorphic rock a hornfels.
With carbonate country rocks, a marble or skarn.
As with most forms of high-T metamorphism, grain sizes usually increase. At the low
confining pressures typically involved, hornfels grains develop little if
any parallelism—indeed, any pre-existing parallelism may be lost in contact
metamorphism. The photo at right shows a distinct
gradient in the contact metamorphism of red Maroon
Formation rocks to green hornfels near Cathedral
Lake, Elk Range.
If hot, mineral-laden fluids emanating from or mobilized by the intrusion
further alter the country rocks, the process is called metasomatism.
Metasomatism is single-handedly responsible for the mineralization of the Colorado
Mineral Belt. Metasomatic changes are usually most pronounced when carbonate
rocks surround silica-rich intrusions, and that's precisely the
combination—silicic Laramide
and mid-Tertiary intrusions placed
within or near the Leadville Limestone—that
set Leadville and Aspen on the road to mining fame. The metasomatic aureole
surrounding an intrusion is usually less extensive than the thermal aureole.
Another form of high-T, low-P granulite-fascies
metamorphism occurs during closure of backarc
basins, which are heated both by arc magmatism
and by hot asthenosphere brought near
by lithospheric thinning beneath the basins. This backarc basin signature
is valuable to geologists investigating collisional
continental crust.
Subduction-related (high-P, low-T) Metamorphism
Sedimentary rocks like graywackes caught up in the accretionary
wedge of a subduction zone and
entrained by the cold sinking slab can quickly reach depths of 20 km or more.
If they're spit back out before they can heat up to temperatures appropriate for
such depths, they reappear as blueschists. (No one knows exactly how blueschists rise again, but
buoyancy relative to surrounding mantle is probably involved.) The amphibole mineral glaucophane provides
the blue cast.
No other known tectonic process can produce the high-P, low-T "blueschist" minerals (lawsonite, glaucophane,
jadeite, pumpellyite) that develop in the subduction environment. Finding blueschist in
the field is a surefire marker of subduction.
Eclogite is another high-P, low-T metamorphic product of subduction
formed at ~60 km by a phase change in the basalt and gabbro making up oceanic
crust. Eclogite's great density and depth of formation make it an important subduction
accelerant. It returns to the surface only under very unusual circumstances.
Submarine (low-P, low-T) Metamorphism
Hydrothermal brines circulating through abundant cracks in the newly-formed
oceanic crust near spreading centers chemically alter the seafloor basalts and
gradually fill the cracks with calcite precipitated directly from the hot
seawater. The resulting green fine-grained igneous rocks, rife with
crisscrossing
white veins, are called greenstones. They find their way onto land in
subduction zone collisions that uplift fragments of oceanic lithosphere or
accretionary wedges. The Early Proterozoic Green
Mountain arc and Farwell-Lester shear zone of northern Colorado record such
collisions during the early assembly of the Colorado
Province.
Cataclastic (high strain) Metamorphism
 |
Granitic mylonite
|
Cataclasis results when rocks around faults or other intense
deformations fracture on very small scales. Reduction in grain size is the most
common signature; metamorphic minerals don't usually form because temperatures
are typically low. At greater depths, mylonites form by recrystallization with
crystal orientations dictated by local stresses or plastic flow. At right is a
1.7 Ga quartz monzonite mylonite exposed in Golden
Gate Canyon State Park in the Front Range.
 |
Homestake SZ
|
Recrystallized mylonites can end up stronger than the undeformed rocks around
them. Vertical fins of tough 1.7 Ga mylonite (right) mark the Homestake shear
zone (SZ) along Homestake Creek in the northern Sawatch Range. The cataclastic foliation
is obvious even at this distance.
Burial (low-P, low-T) Metamorphism
When sediments interlayer with volcanic rocks in a deep sedimentary basin
like the Denver basin, they can reach
temperatures of several hundred degrees. High water pressures foster changes in
mineralogy without disturbing rock textures or fabrics. Zeolites are a common
product of burial metamorphism, but some geologists consider it to be
nothing more than high-grade diagenesis.
Once a Gneiss,
Always a Gneiss
Since
chemical and physical metamorphic reactions rarely take more than days to weeks to complete
in the laboratory, even in solid rock, they have no trouble keeping up with changing
pressure-temperature (PT) conditions in metamorphic environments, where PT
conditions typically vary on timescales
measured in thousand to millions of years.
With rocks that responsive, you might think that they'd also readily revert to their original state if the PT changes were
undone, but that's seldom
the case. Chemical and textural metamorphic alterations are generally irreversible. Once a
shale progrades into a gneiss
(like the Royal Mountain hornblende gneiss at right), stable at, say, 500°C, it can't retrograde back to a shale
on uplift and re-exposure at the surface. It will weather and erode as a gneiss
instead. Granted, the feldspars and ferromagnesian minerals in the gneiss may
well break down into clay redeposited as shale one day, but a direct reversion
to shale can't happen.
Among the many reasons for irreversibility are the general increase
in grain size, the decrease in porosity and the release of volatiles (most notably water and carbon dioxide) common
to many metamorphic alterations. Once
the volatiles escape the rock, there's no turning back—at least not to the
pre-metamorphic mineral assemblage. Retrograde metamorphic minerals may
form in very small quantities (for example, at the margins of higher grade
crystals), but prograde minerals dominate any
metamorphic rock returned to the surface. If the minerals don't revert, the
metamorphic fabrics and texture they create won't change, either.
Footprints
Note that the irreversibility of metamorphic changes means that a metamorphic rock
faithfully records in its mineral assemblage and fabrics the maximum
PT field it's endured. That in turn tells a good bit about where the rock's
been. Geochemists and geophysicists have been busy working out which metamorphic
changes go with which PT fields since the Finnish geologist Eskola first
proposed such associations in 1915. Geologists seeking to unravel metamorphic
terrains make heavy use of such relationships.
Metamorphic Fabrics and Textures
 |
1.7 Ga schist with boudins, Genesee Mountain
|
In the field, metamorphic rocks are often identified as such by their
distinctive fabrics and textures.
What's the difference? Fabrics relate to the alignment of grains, while
textures relate to the distribution of grain sizes and and the normalcy of
grain shapes. Metamorphic
fabrics and textures usually replace similar pre-metamorphic features, like
flow-related sedimentary grain alignments, but they often superimpose in
previously metamorphosed rocks.
Metamorphic Fabrics
 |
Foliation
|
Metamorphic minerals with elongated or platy shapes respond to unequal
stresses by recrystallizing in orientations that reduce strain. That usually means
putting the new shortest grain dimension along the axis of greatest compression.
A grain alignment produced in this way is called a metamorphic fabric—one
of hallmarks of metamorphic rock. If you find a crystalline rock with a fabric,
it's most likely metamorphic, but note that non-metamorphic intrusives can
acquire flow fabrics through magma currents present prior to
freezing.
Fabrics can be dated by noting which independently datable structures (like plutons)
they do and do not involve. Features like wings on porphyroclasts can be used to
determine their sense of motion. Rocks with complex
deformation histories often display several superimposed metamorphic fabrics;
teasing these apart in the field is one of the goals of structural geology.
 |
Schistosity
|
Foliation
arises from the coplanar alignment of platy crystals. Strong foliation involving
small grains results in cleavage—the ability to split a rock in to
highly planar sheets. Schistosity is the alignment of coarse platy or
elongated grains. Schistose rocks can still be broken into small planar flakes,
but not into large sheets. Subsequent stresses can deform the plane of
schistosity, as seen in the porphyroblastic
schist at right from Golden Gate Canyon State Park.
Lineation arises from the alignment of elongate
but not platy grains, or from the intersection of two non-coplanar foliations.
Note that lineations are not just foliations seen on end. They are truly
one-dimensional fabrics, but most lineations turn out to reflect alignments of
elongated grains within a plane of foliation.
[photo coming]
|
Banded gneiss
|
Gneissic banding reflects the segregation of felsic
(light-colored) and mafic (dark-colored) minerals perpendicular to the maximum
compressive stress. The bands may range from millimeters to several meters in
thickness and may be straight or highly contorted, particularly is the rock's
undergone plastic flow.
Metamorphic Textures
Whereas fabrics
relate to grain alignments, textures relate to the distribution of grain
sizes and the normalcy of grain shapes. Metamorphic
processes generally foster larger grains of roughly equal size. A metamorphic
rock with such an equigranular texture is said to be granoblastic.
That said, porphyroblasts,
crystals much larger than average grain size, are not uncommon in
metamorphic rocks, which are then said to be porphyroblastic in texture.
 |
Boudins in 1.7 Ga schist, Genesee Mountain
|
Cataclastic
(high-strain) metamorphism produces mylonites. Under
high-strain conditions, grains of particular minerals (often feldspars) can aggregate into prominent porphyroclasts embedded in a much finer
foliated
matrix.
If the porphyroclasts are eye-shaped, as they often are, they're called augen.
In the field, certain porphyroclast shapes serve as valuable sense-of-shear
indicators in porphyroclastic
metamorphic rocks. Veins may be pinched and pulled into sausage-shaped bodies
called boudins.; the process is called boudinage. In the tortured
schist at right, the light-colored pegmatite veins show
boudinage.
Under ideal conditions, minerals grow into crystals with characteristic
shapes, but when new minerals grow or old ones recrystallize in a real
metamorphic rock, they may or may not be able to assume their ideal shapes. If
they succeed, the rock is said to have an idioblastic texture. If they
fail badly, the texture is xenoblastic (strange). In between are hypidioblastic
textures.
Flow Patterns
Wavy strands and sausage-shaped boudins generated by plastic flow under extreme PT
conditions are seen in migmatites at the brink of wholesale
melting and in ductile banded gneisses. Such features are transitional between
fabrics and textures. The wavy white feldspar strands within the Homestake shear zone
mylonite at right indicate deformation at temperatures near the melting
point; sense of shear can be inferred from the wavy shapes.
For more on metamorphic textures and fabrics see Lynn Fichter's ^Foliated
Textures of Metamorphic Rock.
Metamorphic Minerals, Grades and
Facies
The minerals produced by metamorphism are determined to some extent by parent rock composition but
to a greater extent by the maximum temperature and pressure to
which the minerals equilibrated, with little if any retrograde
alteration on return to the surface. Certain metamorphic
mineral assemblages have thus become associated with certain PT fields. These associations are called facies.
The table below gives an idea of the minerals and PT fields involved; the
temperatures and pressures shown are very rough approximations.
Metamorphic Facies
|
| Facies |
Common Minerals |
Temperature (°C) |
Pressure (kbar) |
| Hornfels |
Biopside, biotite, andalusite |
250-700 |
<2 |
| Greenschist |
Chlorite, epidote, albite |
300-400 |
<6 |
| Amphibolite |
Hornblende, garnet, biotite, sillimanite |
550-650 |
<10 |
| Granulite |
Garnet, plagioclase, kyanite, sillimanite |
650-1200 |
low |
| Blueschist |
Glaucophane, lawsonite, pumpellyite, jadeite,
chlorite |
200-400 |
6+ |
| Eclogite |
Jadeite, garnet, kyanite |
500-700 |
>10 |
Identification of the mineral facies present in a metamorphic outcrop thus
gives an idea of the maximum PT conditions, and to a lesser extent, of the
parent rocks involved. When a rock contains blueschist facies minerals, for
example, it's said to be of blueschist grade, and we cans safely infer it's
spent time in a high-P,
low-T subduction zone. Other facies are less specific with regard to the
tectonic environment but provide important information nonetheless.
Common Metamorphic Rocks
The metamorphics derived from quartz sandstones (quartzites)
and mudstones (slate, phyllite,
schist, gneiss and migmatite)
have
already been discussed in some detail above. Note, however, that schist,
gneiss and migmatite are final
common metamorphic pathways for many parent rocks, not just mudrocks. This section
describes some other common metamorphic rocks. []
Other Common Metamorphic Rocks
|
| Metamorphic Rock |
Parent Rock Examples |
Notes |
| ^Marble,
skarn |
Carbonate country
rocks near intrusions |
- Fine-grained, high-T, low-P product of contact
metamorphism
- Purity determines color and texture
- Skarn is a lower-purity variant containing crystals of
calc-silicate minerals like garnet
|
| ^Hornfels,
granofels |
Silicate country rocks near intrusions |
- Fine-grained, high-T, low-P product of contact
metamorphism
- At the low pressures typically involved, hornfels grains develop or retain little if any
parallelism
- Coarse-grained variant is called granofels.
|
| Greenstone |
Submarine basalt |
- Name refers to any metamorphic rock dominated by chlorite, which
gives the green
- Often, the product of submarine low-P, low-T submarine
metamorphism at spreading centers
|
| ^Greenschist |
Mudstone, mafic |
- Chlorite is the dominant mineral, hence the green color and the name
|
| ^Amphibolite |
Mudstone, mafic |
- Amphibole minerals dominate
|
| Granulite |
Deep sediments in closing backarc
basins at collisional subduction
zones |
- Pyroxene is the dominant mineral
- Granoblastic rock composed of quartz, feldspar and lesser
pyroxene or garnet
- Presence implies high-T, low-P conditions
|
| ^Blueschist |
Subducted accretionary
wedge materials |
- Presence of blueschist minerals implies subduction to high-P, low-T
environment (6+ kbar)
- Blue color comes from glaucophane
|
| ^Eclogite |
Basalt and gabbro in subducting
slabs; also, in underplated depleted mantle source regions beneath
fossil magmatic
arcs like the Sierra Nevada |
- Formed only at depths >~ 60 km and pressures > 10 kbar
- In situ exposures are rare; found also in mantle xenoliths
- Formation of eclogite in falling slabs makes them denser, thus speeding
subduction
|
| ^Serpentinite |
Ultramafic mantle peridotite just above subducting
slabs |
|
| Mylonite |
Any |
- Product of cataclastic
metamorphism
- Recrystallization parallel to local stresses ±
signs of plastic flow
- Ultramylonites are even more thoroughly fractured
|
Table Note: The external links lead to illustrated descriptions of
metamorphic rocks at Lynn Fichter's Metamorphic
Rocks site.
Metamorphic Map Units
On the Geologic Highway Map of Colorado, the 1.7
Ga metasediments and metavolcanics making up much of the Precambrian
basement are mapped as "Xm" in medium gray, while the 0.9-1.4 Ga
metasediments of the Uinta Group and the Uncompahghre
Formation appear in medium gray with wavy lines under the label "Ym".
The craggy hornfels outcrops of the southern Elk Range
are lumped with the Maroon Formation
under the light blue "PP" unit.
Top
Page Index
Rock Formations and Sequences
In this section, we'll discuss some of the
basics of stratigraphy, the science of deciphering stacks of sedimentary
rocks.
What Are Rock Formations and Sequences, And What Good Are They?
 |
The distinctive Morrison Formation,
seen here in the slope of the Grand Monocline near Grand Junction, is
easily recognized statewide by its gray, purple and green color scheme and
its slope-forming habit.
|
A formation is a set of rock layers that share an age range and one or
more distinctive physical characteristics, can be followed for some distance as
a group, and can be drawn on an outcrop map. The more recognizable layers within
a formation are often called members or units. Since formations
deal in layered rocks, we usually think of sedimentary
formations, but it makes just as much sense to speak of volcanic
formations in some settings, and minor ash units are
tolerated in predominantly sedimentary formations like the Cretaceous Pierre
Shale. At right, the Grand Monocline at Colorado
National Monument exposes the distinctive and widespread Morrison
Formation.
The mappability criterion is an important one, for the true extent and
distribution of a sedimentary package yields important clues about its
provenance. Many of the synorogenic
sedimentary strata of the upper Denver Basin vary too much in thickness,
lateral extent or even sediment type (fascies) to be truly mappable. When Robert
Raynolds undertook to reorganize the confusing welter of named strata (some
already with formation status) into mappable units, he ended up with only two
— an older D1 sequence and a younger D2 sequence
separated by an easily recognizable unconformity
and paleosol. Unconformities also demarcate the bottom of the D1 and the top of
the D2.
Formation Names
Formations consisting mostly of a single rock type receive a place name
indicating the type locality (the place where the formation was first
recognized) followed by the name of the dominant rock, as in "Leadville
Limestone". Formations featuring more than one significant rock type
get a place name followed by "formation"—hence the "Morrison
Formation", which contains claystone, sandstone and minor limestone
members. Formations found together over large areas may be lumped into a group.
The Dakota Group of eastern Colorado
subsumes an upper South Platte
Sandstone and a lower Lytle Formation
member.
Formations are often divided by terms like upper and lower to
reflect the relative positions of included rock units as they appear in the
field. The upper units are usually younger than the lower units, but a
word of caution is in order here: Things can get upside down now and then. In a
sedimentary formation caught up in an overturned anticline, the lowest units
will be younger than the middle strata.
Formation Boundaries
Upper and lower formation boundaries may be fairly distinct on paper, but
they aren't always conspicuous in the field. For instance, the lower sands of
the early Cretaceous Lytle Formation are
distinguished from
the upper sands of the late Jurassic Morrison Formation only by
the presence of small chert pebbles weathered out of Paleozoic
limestones and dolomites exposed far to the west. You'll have to get pretty
close to tell them apart.
 |
Gently-dipping late Devonian Elbert sandstones rest unconformably on
nearly vertical Uncompahgre Formation
quartzites in Box Canyon near Ouray
|
Unconformities (depositional gaps wherein one formation accumulates on
the eroded surface of another) usually provide the sharpest and most conspicuous
formation boundaries, especially if the strata change dip across the gap — in
which case it's an angular unconformity. Proponents of sequence
stratigraphy would argue that unconformities are also the most useful of
stratigraphic boundaries.
At right, gently dipping late Devonian sandstone of the Elbert
Formation rests unconformably on nearly vertical 1.6 Ga Uncompahgre
Formation quartzites above Box Canyon Falls just south of Ouray off US 550.
This famous angular unconformity draws geologists from all over the world, but
the volunteer working the visitors' center knew absolutely nothing about it. Why
am I not surprised?
Lateral Variations and Terminations
Formations have a nasty habit of changing laterally in devious ways.
Eventually, they all die out against the margins of their receiving basins, some
sooner than others. In one form or another, the upper Cretaceous Dakota
Group has a 9-state distribution, but the synorogenic
Laramide sediments east of the Front Range cover at most an 80 x 150 km area
along the mountain front. They get thicker here and thinner there and pinch out altogether at
many points in between.
 |
Late Cretaceous Mancos shale
ends locally and abruptly against Precambrian metasediments along the
Laramide Cimarron fault south of Black
Canyon, here seen from eastbound US 50 just east of Cimarron
|
Formations may have a distinct lateral margin in one location (e.g., against an
erosional surface or a fault, as in the photo at right) and interfinger with laterally equivalent formations in messy ways
elsewhere,
or they may just up and change names at some arbitrary and usually unspecified
boundary for reasons that are almost always more historical (in the history of
science sense) than logical. Thus we're left with a Dakota Sandstone in western
Colorado depositionally equivalent to the upper South
Platte Sandstone of the Dakota Group of eastern Colorado. Formations may
also contain lenses of minor or even major units here but not there. What a
headache!
Formations As Mental Tools
Such is the inherently untidy nature of sedimentary and volcanic transport
and deposition in the real world. Throw in some
faulting and a small uplift or intrusion here and there, and you can easily end up with a huge
mess in the field.
When dealing with rocks and stories about rocks, one must bear in mind at all
times that "formations" are really nothing more than hopeful human
constructs foisted upon a planet with no apparent limit to its taste for
complexity. Nevertheless, formations give us a convenient and often useful if flawed way to speak of and
think about earth materials with common histories. They help us chunk certain
details long enough to work out a bigger picture of things in time and space.
That's how they earn their keep.
Sequence Stratigraphy
Recent advances in stratigraphy now favor sequences over formations as the
logical and map units of choice for many purposes, including the reconstruction
of paleogeographic settings and the events that punctuate them. A sequence
is a stack of related sediments bounded above and below by an unconformity or an
equivalent updip conformity. Event-oriented sequences mark distinct depositional
regimes punctuated by tectonic rearrangements of depositional basins, source
areas or both. Sequence stratigraphy's seemingly endless terminology can be
off-putting at first, but it's carefully crafted to bring the full power of ^Steno's
principles (first formulated in the late 1600s) and ^Walther's
law to bear in the pursuit of depositional origins. The table below
summarizes these tried and true maxims of sedimentology.
The Foundations of Stratigraphy—
Steno's Principles and Walther's Law
|
| Title |
Statement |
Key Implications |
Colorado Examples |
Principle of
superposition
|
In the absence of severe deformation, the
youngest strata lie on top and lower strata are successively older
|
The order of undeformed strata parallels
the order of events leading to their deposition
When an older stratum overlies a younger one, or the bottom of a
stratum is now on top, the stack has been overturned by subsequent strong
deformation
|
 |
Steeply west-diipping overturned Laramie Fm sandstone ridge at Golden;
exposed triceratops track depressions nearby now poke out to the
west
|
|
Principle of
original horizontality
|
Due to gravity, fluid-borne sediments must
layer horizontally when first deposited
|
Strata that are no longer horizontal have
been deformed after deposition
Outcropping strata sharing the same non-horizontal dip probably occupy
one limb of a larger fold
When strata rest on older strata dipping at a different angle (across
an angular unconformity), a mountain-building event has intervened
|
 |
Gently-dipping late Devonian Elbert sandstones rest unconformably on
nearly vertical Uncompahgre
Formation quartzites above Box Canyon Falls near Ouray
|
|
| Principle of
original lateral continuity |
Deposits originally extend in all directions
to either taper away or otherwise end against the receiving basin's
margins |
If a reliably identifiable stratum is now
discontinuous, the missing sections have been eroded, folded or faulted
away [] |
 |
Dakota sandstone caps the
east wall of the lower Animas canyon just above Durango; between here and
the Dakota Hogback east of the
Front Range, erosion has stripped the once continuous Dakota from several
Laramide uplift summits
|
|
Law of
lateral succession
|
As depositional basins evolve (subside,
tilt, invert), their margins migrate over time; thus, vertically stacked
strata also succeeded each other laterally
|
Originally lateral successions (e.g.,
lagoon, beach, offshore bar, shallow marine and deep marine sediments) can
pave over large areas with vertical sequences of the same strata in the
same order
|
|
Raynolds' D1 sequence of synorogenic
strata east of the Front Range tells of the erosional unroofing of a rising
central Front Range basement block covered at the time (~67-68 Ma, in the
Paleocene) with Laramide-aged andesitic volcanics. As this depositional event
came to a close, erosion attacking the top of the D1 created an unconformity.
Some 8 Ma later, the arrival of the D2 sequence heralded the rise and
accelerated erosion of the Pikes Peak and Rampart Range segments of the Front
Range to the south. These new source areas shed feldspar-rich (arkose) gravels,
sands and muds derived mostly from the Pikes Peak granite into the same basin
that trapped the D1 sequence.
Here's a decent ^primer on
sequence stratigraphy. While most sequence stratigraphy sites focus on
marine sediments, it's no less applicable and profitable in terrigenous
environments.
Rock Mechanics
The idea of bending and flowing solid rock flies in the face of common
experience, but once you take it to heart, the Colorado landscape becomes much
easier to understand. Here, we'll try to
make the concept a bit more believable.
Butter
Take a cold stick of butter from the refrigerator. It's solid yet plastic.
Press on it with the back of a teaspoon, and it flows slowly out the way but
retains its deformed shape when you remove the spoon. Whack the other end of the
stick with the spoon. Note that the butter flows more when you apply the
deforming force over time. Now scrape the stick with the edge of your spoon and
note how it erodes.
Next, heat the stick in the microwave until it visibly softens short of
melting. Let it cool a minute or so, then press and scrape again with the spoon.
The butter flows and erodes much more readily but still retains its shape when
you remove the spoon. (If it doesn't, you partially melted it.) After a
night in the freezer, the butter would hardly yield
to your spoon press or scrape, but you might be able to break off a piece with a
really good whack.
Believe it or not, rock behaves a lot like butter. Sure, the forces,
temperatures and timescales involved are orders of magnitude greater with rock,
but the basic features of its plastic behavior are much the same. Its plasticity
increases dramatically with temperature, especially near its melting point. At
low temperatures, like those at the earth's surface, it becomes brittle enough
that it would more likely fracture than flow if deformed rapidly.
Just Add Time
 Despite
clear geologic evidence to the contrary, our usually reliable day-to-day sensibilities
maintain that
rock neither bends nor wears. The disconnect occurs because, unlike butter,
solid rock reveals its malleability only over time scales
very long compared to human events—typically over spans of tens of thousands if
not millions of years. We can barely conceive of such intervals, but given that
kind of time, rock will yield to flowing water and
will bend or flow rather than break. Despite
clear geologic evidence to the contrary, our usually reliable day-to-day sensibilities
maintain that
rock neither bends nor wears. The disconnect occurs because, unlike butter,
solid rock reveals its malleability only over time scales
very long compared to human events—typically over spans of tens of thousands if
not millions of years. We can barely conceive of such intervals, but given that
kind of time, rock will yield to flowing water and
will bend or flow rather than break.
Even if it's beyond our ken, the planet's had ample time for the bending and
wearing of rock—hundreds to thousands of times over, in fact. Cold sedimentary
strata near the surface can be folded without breaking in under a million years.
It takes a properly motivated mountain river only a few times longer to cut a
2,700' deep hard rock canyon like the Black
Canyon of the Gunnison. It took only 50 Ma to uplift and erode away the Ancestral
Rocky Mountains and to bury half the state in their remains. You could have
done that 40 times just since the onset of plate tectonics at ~2.0 Ga.
Heat and Pressure Help
 |
Hornsilver mylonite
|
Under great confining pressures, or at depths
where temperatures along the geothermal
gradient where they reach a significant fraction of their melting points (typically 10-15 km),
rocks that are quite brittle at the surface become sufficiently plastic to deform
without fracture at rates comparable to
the rate at which fingernails grow ( ~10 mm/yr, or 10 km/Ma). Granted, that makes molasses look
downright mercurial, but then relative viscosity is the whole idea here.
My piano tuner's job security rests on the fact that properly tuned piano wire flows
(stretches) at a similar rate and falls out of tune in a matter of months. Over
a few Ma, fingernail speed is plenty fast enough to fold great thicknesses of
sedimentary rock over the east edge of the relatively brittle Front Range
basement block, as in the photo at right. Over 50 Ma, an entire mountain range
like the Ancestral Rockies can rise up and vanish.
At right is a 1.7 Ga mylonite exposed at Hornsilver Ridge along the
Homestake shear zone in the northern Sawatch Range. This specimen deformed some 10-14 km below the
surface. The wavy pink feldspar bands are clear signs of
highly plastic flow. Mylonites
develop their metamorphic textures at depth near
moving faults, which add frictional heating to the mix. This one came close to
melting but still deformed as a solid.
 |
Idaho Springs-Ralston shear zone schist
|
The dark Z-shaped inclusions in the final image at right are not the work of
Zorro. They are recumbent isoclinal folds in 1.7 Ga schist from the Idaho
Springs-Ralston shear zone at Golden Gate Canyon State Park. Distinct folds
like these can only form in solid rock with considerable plasticity. Molten rock
would never preserve such structures.
Brittle-Ductile Transitions
Local geothermal and pressure
gradients interact with the materials in the local rock column to establish
one or more brittle-ductile transitions (BDTs) within each distinct column in
the crust. Rocks above a BDT fracture when applied
stresses and resultant strains exceed certain limits, while rocks below deform
in a plastic manner with little if any fracturing. Anomalously high temperatures
or pressures at depth generally raise the BTD, which, for example, is much
shallower than usual in "hot orogens" like magmatic
arcs and backarc basins. Similarly, a
cold slab of oceanic crust falling
into the upper mantle drags the BTD
down with it until it warms enough to lose all brittle behavior, or to ambient
temperature, whichever occurs first.
The primary BDT within the basement defines the boundary between the upper
and lower crust, but the cover may
develop its own secondary BDT above the basement. Consider a typical
monolithic Rocky Mountain basement block
bounded by steep faults inherited from pre-Laramide tectonic events and now
mantled by ~12,000' of flat-lying late
Pennsylvanian to Mesozoic
sedimentary cover accumulated during and since
the rise of the Ancestral Rocky Mountains at
~300 Ma. The time is the latest Cretaceous, say ~70 Ma, and the block has just
begun to rise along its reactivated
(and now reverse) bounding faults at the onset of the Laramide
orogeny. Due to the great thickness of the cover, high confining
(lithostatic) pressures establish a secondary BDT somewhere above the
basement-cover contact, while the primary basement BDT remains well below it. If
a block margin above the basement BDT happens to fail as the block rides up and
over adjacent blocks, it will break up in a brittle manner, just as the Rampart
Range broke off the southeast margin of the Front Range. In contrast, the cover
layers below the secondary BDT will drape over broken block margins like so much
"melted cheese" on a chopped potato. Additional examples of ductile
cover deformation over brittle basement fragmentation occur all along the east
margin of the Front Range north of Boulder.
Acknowledgment: Thanks to Vince Edwards for introducing me to this
compelling model of range-front deformation.
Top
Page Index
References
In addition to the home page references, this
article relies on the following sources, in alphabetical order by first author:
-
Ehlers EG, Blatt H,
1982, Petrology: Igneous, Sedimentary and Metamorphic, W. H. Freeman and
Company, San Francisco
-
Fichter LS, 2000, ^Introduction
to Sedimentary Rocks, ^Introduction
to Igneous Rocks, ^A
Very Simple Classification for Metamorphic Rocks
-
GTR Mapping, 1998, Colorado Geologic Highway
Map, 1991 revision, scale 1:1,000,000 (available at ^MAPSCO, Denver, CO)
-
Hamilton WB, 1989,
"Crustal geologic processes of the United States", in Pakiser LC
and Mooney WD, eds., Geophysical framework of the continental United
States, Geological Society of America Memoir 172, p. 743-781.
-
Johnson KR and Reynolds RG, 2001, Ancient
Denvers—Scenes from the Past 300 Million Years of the Colorado Front Range,
Denver Museum of Nature and Science
-
McCaffrey R, Abers GA, 1991, Orogeny in
arc-continent collision: The Banda arc and western New Guinea, Geology
v. 19, p. 563-566 (June)
-
National Geographic
Society, 1998, Physical
Earth (insert map)
-
Railsback LB, 2003,
An earth scientist's periodic table of elements and their ions, Geology
v. 31, p. 737-740 (September)
-
Scott RB et al., 2001, Geologic Map of the
Colorado National Monument and Adjacent Areas, Mesa County, Colorado,
scale 1:24:000,
Geologic Investigations Series I-2740, USGS (available at the Colorado
National Monument Visitor's Center)
-
Taylor AM, 1992, Guide Book to the Geology of
Red Rocks Park and Vicinity, Cataract Lode Mining Company
-
Taylor AM, 1999, Guide to the Geology of
Colorado, Cataract Lode Mining Company
-
Tweeto, Ogden, 1979, Geologic
Map of Colorado, scale 1:500,000 (available at ^MAPSCO,
Denver, CO)
-
US
Geological Survey Photo Glossary of Volcano Terms
-
www.webmineral.com
gallery of mineral specimens
Top
Page Index
|